INTRODUCTION
Irrational use of antibiotics by healthcare practitioners is a major cause of antimicrobial resistance (AMR) (Solomon and Oliver, 2014). Rising rates of AMR are reported worldwide and associated with increased morbidity, mortality, and costs of healthcare (Founou et al., 2017). By 2050, AMR is expected to cause 10 million deaths yearly if proper actions are not taken (O’Neill, 2014). The worst impact of AMR will be in the developing world due to poor healthcare systems and limited resources (Founou et al., 2017).
Irrational use of antibiotics in hospitals can take different forms. This includes, but is not limited to, overprescribing of antibiotics, unnecessarily long durations of treatment, the use of broad-spectrum antibiotics without clear indications, and wrong dosing (Ventola, 2015). Therefore, stewardship strategies should be seriously taken to enhance the responsible use of antibiotics (Dyar et al., 2017). Several strategies have been suggested, the top of which is adopting and effectively implementing guidelines for antibiotic use, whether in the treatment or in the prevention of infectious diseases (Resman, 2020).
Healthcare-associated infections (HCAIs) are acquired by patients receiving healthcare in hospitals. HCAIs are common, need additional medical interventions, and can lead to prolonged hospital stay with increased expenditures (Haque et al., 2017; Napolitano et al., 2013). In developing countries, 10% of hospitalized patients develop HCAIs, with surgical site infections (SSIs) being the most common among all HCAIs, affecting up to 5.6% of all surgical patients (Allegranzi et al., 2011). Many interventions have been studied to prevent SSIs and were shown to be effective. These include aseptic procedures in the operating room, skilled surgical techniques, postsurgical wound care, and perioperative antimicrobial prophylaxis (PAP) (Napolitano et al., 2013). The use of PAP decreases bacterial load in the wound leading to an 80% decrease in the likelihood of SSIs, regardless of surgical procedure type or wound contamination (Bowater et al., 2009). Yet, PAP is not needed for all surgical procedures and should only be applied if SSI-related costs and morbidity exceed those associated with antibiotic prophylaxis (Herawati et al., 2019). Evidence-based guidelines were established to augment the rational use of PAP, stating when it is indicated, proper timing of administration, duration of use, and suitable antibiotics for this purpose (Bratzler et al., 2013). Despite this, evidence from developing countries shows that guidelines are not always followed. Reports from Jordan (Al-Momany et al., 2009), Saudi Arabia (Ahmed et al., 2022; Tolba et al., 2018), and Qatar (Abdel-Aziz et al., 2013) have found a wide gap between the actual practice of PAP and clinical practice guidelines. PAP is overprescribed, used in subtherapeutic doses, for long durations, and antibiotics used are not those recommended by the guidelines. This malpractice adds to the problem of antibiotic misuse and may increase the costs and risks of both adverse drug reactions and AMR.
Limited information about PAP prescribing trends and adherence to practice guidelines in Palestine (West Bank/Gaza Strip) is available. Only one paper has been published on this topic from the West Bank, showing a very poor adherence rate (2%) (Musmar et al., 2014). No studies, however, have been conducted in Gaza Strip hospitals. Hence, this study aimed to assess the adherence of PAP practice in Gaza Strip hospitals to the guidelines of the American Society of Health-System Pharmacists (ASHP) for antimicrobial surgical prophylaxis.
METHODS
Setting and design
This is a cross-sectional study to assess PAP practice in Gaza Strip hospitals. It was carried out in surgical departments (general and orthopedic) at the following general hospitals: Al-Shifa Medical Complex (SMC), Gaza Governorate; Nasser Medical Complex (NMC), Khan Younis Governorate; and the European Gaza Hospital (EGH), Rafah Governorate. Data were collected over a six-month period during the year 2017.
Patients
The minimum sample size required to power the analysis in this study is 263. It was calculated based on the following assumptions: rate of the appropriate use of PAP of approximately 22%, confidence interval (CI) of 95%, and tolerable level of type 1 error of 5%. Yet, we included all eligible patients undergoing general surgical or orthopedic surgical procedures at the three hospitals over the 6-month study period. Patients from both genders undergoing elective clean or clean-contaminated surgeries (CDC, 2022) were eligible. We excluded those with infection manifestations after surgery and those undergoing gastroesophageal reflux disease procedures (Nissen fundoplication) and endoscopic retrograde cholangiopancreatography, as there is no consensus on the use of PAP for those procedures. In addition, patients undergoing minor surgeries such as gastroscopy or colonoscopy, cancer patients, and those with unclear or incomplete medical records were also excluded. During the study period, 1,514 patients underwent orthopedic or general surgical procedures, of whom only 444 patients were eligible.
Data collection
Relevant data were collected and documented on a standardized case report form. Two data collection methods were utilized: chart review method and direct observation method. The medical chart of each patient was reviewed, and the following data were collected: patient care ward, age, gender, weight, allergy history, American Society of Anesthesiologist (ASA) score, characteristics of surgical procedure, prescribed antibiotics for PAP, dose, administration route, duration, and the number of doses. The observation method was used to report the timing of administration of the first antibiotic dose in relation to incision time and which health professional administered it.
Analysis of PAP use
Appropriateness of PAP was assessed by comparing it to the joint guidelines of the Infectious Diseases Society of America, the ASHP, the Society for Healthcare Epidemiology of America, and the Surgical Infection Society (Bratzler et al., 2013). To simplify, we referred to those guidelines as ASHP guidelines. The following aspects of PAP were assessed: a) indication, b) antibiotic selection, c) duration of use, d) dosing, and e) timing of administration of the first PAP dose (Table 1).
PAP practice was judged adherent to guidelines if all aspects followed the recommendations of the guidelines. For antibiotics given without indication, the parameters of selection, dosing, the timing of administration of first dose, and duration were not assessed. The overall adherence rate to guidelines was calculated by summing up all patients who were eligible for PAP and were delivered the correct antibiotic at the correct dose, the timing of administration of first dose, and duration to those patients who were not eligible for PAP and not delivered it divided by the total number of patients. The indication adherence rate was calculated by summing up all patients who were eligible for PAP and were delivered it to all patients not eligible for PAP and not delivered it divided by the total number of patients. Finally, adherence rate for each other aspect of PAP use (selection of the antibiotic, dosing, duration of use, and timing of administration of the first dose) was calculated by dividing the number of patients delivered PAP correctly with respect to that specific aspect by the number of patients delivered PAP when it was indicated.
 | Table 1. Criteria to assess adherence of perioperative antimicrobial prophylaxis to guidelines. [Click here to view] |
Data entry and analysis
Data were analyzed using the SPSS version 24 program. Continuous variables were presented as means ± SD and compared using the ANOVA test, while categorical variables were presented as percentages and compared using the chi-square test. The chi-square test was utilized to study the relationship between adherence to PAP guidelines (dependent variable) and factors (independent variables) such as patient care ward, specialization of provider administering the antibiotics, and characteristics of surgery (type, grade, class, length, and type of anesthesia). The results were considered statistically significant if p-values were ≤ 0.05. Variables shown to be significant in univariate analysis at p ≤ 0.05 were entered into a multivariable logistic regression model, and results were expressed as odds ratio (OR) with 95% CI and p-values.
RESULTS
Four hundred and forty four patients were eligible and included in the analysis (233 from EGH, 130 from SMC, and 81 from NMC). Patient and surgical procedure characteristics are shown in Table 2. Four hundred and twenty one patients (94.8%) received PAP, most of whom (73.9%) received one antibiotic. In EGH and NMC, cefazolin was the most commonly used antibiotic while ceftriaxone was the most commonly used antibiotic in SMC. PAP was administered by nurses most of the time in both EGH and NMC, while anesthesiologists administered PAP more commonly in SMC. PAP utilization patterns are shown in Table 3.
Overall adherence to current PAP practice and adherence of each parameter of PAP use to guidelines are detailed in Table 4. Overall adherence was fulfilled in 33 cases only (7.4%), while the indication adherence rate was 70.7%. Significant differences were observed among hospitals, with NMC showing better overall adherence and indication adherence rates than EGH and SMC.
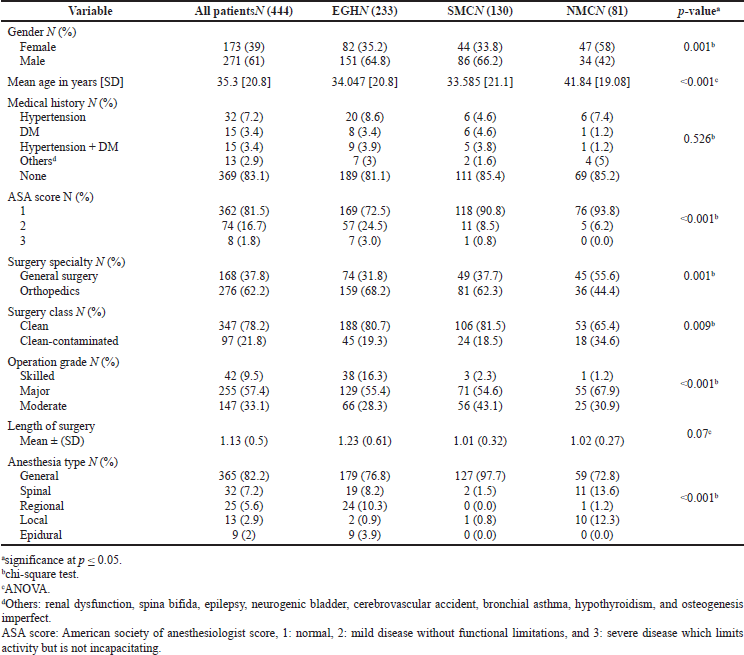 | Table 2. Characteristics of patients and surgical procedures. [Click here to view] |
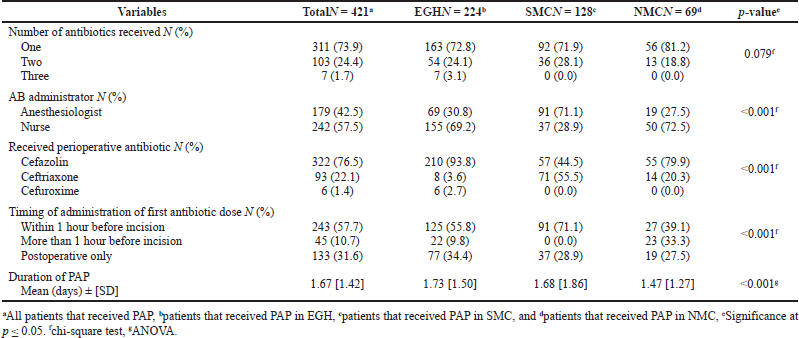 | Table 3. Perioperative antimicrobial prophylaxis utilization patterns. [Click here to view] |
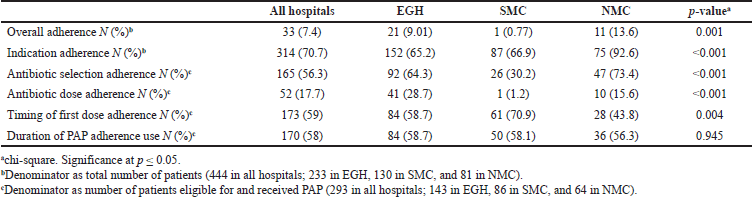 | Table 4. Adherence of perioperative antimicrobial prophylaxis practice to ASHP guidelines. [Click here to view] |
Two hundred and ninety-three patients (66%) were eligible for PAP and were delivered it, and 21 patients (4.7%) were not eligible for PAP and were not delivered it. Antibiotic selection was inappropriate in 43.7% of patients; 24.8% had an unexplained switch from an appropriate or inappropriate preoperative antibiotic to inappropriate antibiotic(s) without microbiologic or clinical indication, and 18.9% were given broad-spectrum antibiotics.
The dose was appropriate in only 17.7% of patients, while the dose was low in 82.3% of patients. Timing of administration of the first dose was appropriate in 59% of patients, but 29.7% of patients received the first dose by the end of the surgical procedure, and 11.2% received it more than 1 hour before the incision. Duration of PAP use followed the guidelines in only 58% of patients. Significant differences were shown among hospitals in both selection and dosing adherence rates, where SMC had a lower adherence rate than EGC and NMC, as well as in the time of administration adherence rates, where SMC had better adherence than EGC and NMC.
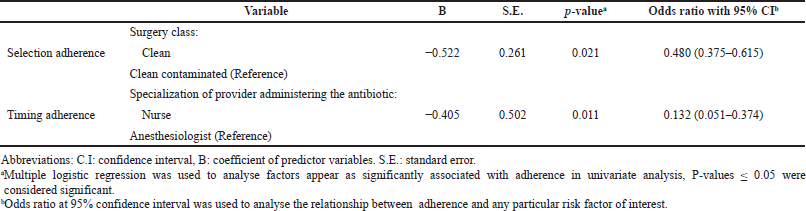
Patient care ward and surgery characteristics were tested for possible association with selection adherence, dosing adherence, and duration adherence to guidelines. The same factors, in addition to the specialization of providers administering the antibiotics, were tested for association with the timing of first-dose adherence. Table 5 presents factors that showed significant association with adherence based on univariate analysis. Multiple logistic regression analysis showed that surgery class significantly and independently affects antibiotic selection (Table 6). Patients who underwent clean surgeries were less likely to be given the recommended antibiotic than those who underwent clean-contaminated surgeries (OR 0.480, 95% CI 0.375–0.615, p-value 0.021). Specialization of providers administering the antibiotics significantly affects adherence to the proper timing of first antibiotic dose administration. Nurses were less likely to adhere to the right timing than anesthesiologists (OR 0.132, 95% CI 0.051–0.374, p-value 0.011). None of the tested independent variables were shown to be associated with dosing or duration adherence rates.
DISCUSSION
A significant gap between clinical guidelines for PAP use and current PAP practice in hospitals in Gaza Strip, Palestine, was shown in this study, with an overall adherence rate of only 7.4%. An earlier study from the West Bank, Palestine, reported a 2% overall adherence rate (Musmar et al., 2014). Poor adherence to guidelines of surgical antibiotic prophylaxis is a common finding shared by many studies from developing countries, with overall adherence rates ranging from 0% to 15.3% (Al-Momany et al., 2009; Kaya et al., 2016; Tolba et al., 2018). Yet, studies from the European Union countries reported higher adherence rates of 71% to 85% (Hohmann et al., 2012; Pittalis et al., 2013). In this study, written guidelines for PAP practice were not available in any of the investigated hospitals. Therefore, PAP practice was based on each clinician’s own knowledge and judgment. This may explain the very low adherence rate to guidelines. Earlier studies have suggested that “standardization,” by adopting PAP international guidelines or by establishing local PAP protocols, is essential to improve PAP practice (Abdel-Aziz et al., 2013; Al-Momany et al., 2009). Another important observation in the study is the complete absence of clinical pharmacy services in areas of surgery in the studied hospitals. Pharmacists can improve PAP practice by developing and controlling adherence to guidelines, monitoring dosing, timing, and choice of antibiotics, and educating other operating room personnel and providing them with accurate drug information (ASHP, 1999). A great body of evidence has demonstrated significant improvements in PAP practice with favorable clinical and economic outcomes by clinical pharmacists’ services and interventions (Telfah et al., 2015; Zhang et al., 2014; Zhou et al., 2016).
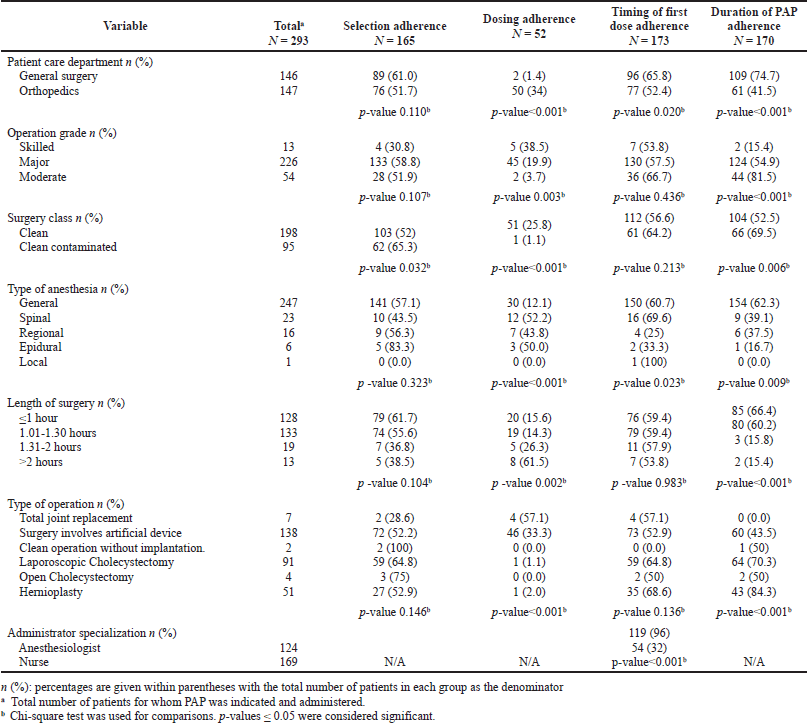 | Table 5. Factors associated with adherence to perioperative antimicrobial prophylaxis guidelines. [Click here to view] |
 | Table 6. Multiple logistic regression analysis of factors associated with adherence to perioperative antimicrobial prophylaxis guidelines. [Click here to view] |
In this study, an indication adherence rate of 70.7% was comparable to that from Italy (70.3%) (Durando et al., 2012) but lower than those observed in studies from the USA (87.1%) (Wright et al., 2013) and France (85%) (Malavaud et al., 2008). In our study, PAP was not indicated but administered in 28.8% of cases and was used for longer than the recommended duration in 42% of cases. Such an “overuse” of antibiotics is shared with other studies from Jordan (Al-Momany et al., 2009), Qatar (Abdel-Aziz et al., 2013), and Palestine (Musmar et al., 2014), where PAP was used for longer durations than recommended in 58.9%, 59.3%, and 68.2% of cases, respectively. Concerns regarding the inadequacy of nonpharmacological preoperative and intraoperative measures used in our hospitals to reduce infections and poor patient wound care at hospital wards and after discharge, added to the belief in the safety of the used antibiotics, may have contributed to such an overuse of PAP. Overuse and prolonged use of PAP can increase risks of adverse effects, promote AMR, and accelerate healthcare costs (Bratzler et al., 2013; McDonnell Norms Group, 2008). Prolonged PAP use was associated with the emergence of resistant organisms, as demonstrated early by Burnett et al. (1980). In their study, cephalothin was used for prophylaxis in hip fracture repair procedures. After 72 hours of use, cephalothin-resistant pathogens were found in 42% of all positive cultures.
Interestingly, while overuse of PAP was reported in this study, dosing was below that recommended by guidelines in the majority of patients (adherence rate of only 17.7%). For example, the dose of cefazolin frequently used for adult surgical patients was 1 g, though the recommended adult dose is 2 g or 3 g for those weighing <120 kg or ≥120 kg, respectively. These findings are consistent with a Jordanian study that reported a 27.9% dosing adherence rate (Al-Momany et al., 2009). As in this study, all incorrect doses were lower than the recommended. This may increase the risk of SSIs, as shown by Crawford et al. 2016). In their study, patients receiving 1 g of cefazolin were more likely to develop SSIs (13.2%) than those not receiving PAP (4.0%, p < 0.0001).
Antibiotic selection in this study was appropriate in only 56.3% of patients, with increased use of third-generation cephalosporins instead of the recommended “cefazolin.” The routine use of third-generation cephalosporins or broad-spectrum combinations is not recommended for SSI prophylaxis due to lower activity against staphylococci than cefazolin, increased risk of inducing AMR, and higher costs (Zhang et al., 2014). Indeed, there is a misbelief among healthcare practitioners in our country that broad-spectrum antibiotics are more effective than narrower-spectrum ones. This, along with the absence of effective antibiotic stewardship strategies, may explain the increased use of broad-spectrum antibiotics, as shown earlier in studies from the Gaza Strip (Al Laham, 2012) and the West Bank (Musmar et al., 2014). Antibiotic selection in this study was more appropriate in patients who underwent clean-contaminated surgeries than in those with clean surgeries. There are many antibiotic options for clean-contaminated surgeries, as recommended by the guidelines, including ceftriaxone, which is preferred by many physicians due to its broad-spectrum activity, as discussed above. Yet, for clean surgeries, the choice is restricted to cefazolin only (Bratzler et al., 2013).
Administration of PAP should take place before contamination occurs. Therefore, ASHP guidelines recommend administering PAP 30–60 min prior to incision to provide sufficient antibiotic concentrations at the time of incision and throughout the procedure (Bratzler et al., 2013). Poor adherence to proper timing of the first antibiotic dose (59%) was observed in this study. Similarly, an earlier study from Palestine (Musmar et al., 2014) reported a 59.8% timing of first dose adherence rate. Better timing adherence rates, however, were documented by studies from Greece (100%) (Tourmousoglou et al., 2008), Jordan (99.1%) (Al-Momany et al., 2009), and the USA (95%) (Goede et al., 2013). A common observation in those studies is that anesthesiologists, not nurses, were responsible for PAP administration. Yet, in our study, PAP was administered by either anesthesiologists or nurses. Further analysis of data in our study found that anesthesiologists were more adherent to the timing of the first antibiotic dose administration than nurses. This was also observed by Musmar et al. (2014), and they explain this by the fact that the skills and training of anesthesiologists—due to their specialty—are related mainly to surgery. Nurses, on the other hand, have a broader scope of patient care duties in hospital wards (Musmar et al., 2014). In their study, Kanter et al. (2006) shifted the responsibility of administering PAP from the nurses to the anesthesiologists, which resulted in a significant increase in timing adherence rates from 11% to 91% (Kanter et al., 2006).
Finally, the strengths and weaknesses of this study are worth mentioning. This study is the first to investigate PAP practice and to give an estimation of PAP appropriateness in Gaza Strip hospitals. It highlighted areas of weakness that need extensive improvements. This study, however, was conducted in the major government hospitals in Gaza, Khan Younis, and Rafah Governorates. Considering that there are many nongovernmental hospitals that serve a substantial number of patients in the Gaza Strip, the generalization of our results cannot be justified.
CONCLUSION
This study highlighted the problem of poor PAP practice in our hospitals. Inappropriateness was observed in all aspects of PAP use, particularly dosing. Strategies that were shown to improve PAP practice elsewhere should be adopted, such as developing and implementing PAP guidelines, as well as involving clinical pharmacists in surgical patients’ care. We also suggest that the responsibility of administering PAP should be assigned to anesthesiologists for better timing of the first PAP dose.
AUTHORS’ CONTRIBUTIONS
Hala Z.I Alagha and Manal Shehata Zourab contributed to the study conception, design, and data collection format preparation. Data collection was performed by Manal Shehata Zourab, and data analysis was performed by Hala Z.I Alagha and Manal Shehata Zourab. Hala Z.I Alagha and Manal Shehata Zourab wrote the original draft and approved the final manuscript.
FINANCIAL SUPPORT
The authors declare they haven’t received any funding for conducting the study or preparing the manuscript.
CONFLICTS OF INTEREST
The authors declare there are no conflicts of interest.
ETHICAL APPROVAL
Approval to conduct the study was first obtained from Al-Azhar University, followed by approval from the Directorate for Human Research, Ministry of Health, Gaza Strip. Following this approval, the institutional review board of each of the study hospitals approved the study.
CONSENT
Informed consent for this type of study was waived as the study was not interventional and did not involve the collection of biological material from patients and collected information was anonymized.
DATA AVAILABILITY
All data generated and analyzed are included in this research article.
PUBLISHER’S NOTE
This journal remains neutral with regard to jurisdictional claims in published institutional affiliation.
REFERENCES
Abdel-Aziz A, El-Menyar A, Al-Thani H, Zarour A, Parchani A, Asim M, El-Enany R, Al-Tamimi H, Latifi R. Adherence of surgeons to antimicrobial prophylaxis guidelines in a tertiary general hospital in a rapidly developing country. Adv Pharmacol Sci, 2013; 2013:842593; doi:10.1155/2013/842593 CrossRef
Ahmed N, Balaha M, Haseeb A, Khan A. Antibiotic usage in surgical prophylaxis: a retrospective study in the surgical ward of a governmental hospital in Riyadh Region. Healthcare (Basel), 2022; 10(2):387. CrossRef
Al Laham NA. Prevalence of bacterial contamination in general operating theaters in selected hospitals in the Gaza Strip, Palestine. J Infect Public Health, 2012; 5(1):43–51. CrossRef
Allegranzi B, Bagheri Nejad S, Combescure C, Graafmans W, Attar H, Donaldson L, Pittet D. Burden of endemic health-care-associated infection in developing countries: systematic review and meta-analysis. Lancet, 2011; 377(9761):228–41. CrossRef
Al-Momany NH, Al-Bakri AG, Makahleh ZM, Wazaify MM. Adherence to international antimicrobial prophylaxis guidelines in cardiac surgery: a Jordanian study demonstrates need for quality improvement. J Manag Care Pharm, 2009; 15(3):262–71. CrossRef
ASHP guidelines on surgery and anesthesiology pharmaceutical services. American society of health-system pharmacists. Am J Health Syst Pharm, 1999; 56(9):887–95. CrossRef
Bowater RJ, Stirling SA, Lilford RJ. Is antibiotic prophylaxis in surgery a generally effective intervention? testing a generic hypothesis over a set of meta-analyses. Ann Surg, 2009; 249(4):551–6. CrossRef
Bratzler DW, Dellinger EP, Olsen KM, Perl TM, Auwaerter PG, Bolon MK, Fish DN, Napolitano LM, Sawyer RG, Slain D, Steinberg JP, Weinstein RA. Clinical practice guidelines for antimicrobial prophylaxis in surgery. Am J Health Syst Pharm, 2013; 70(3):195–283. CrossRef
Burnett JW, Gustilo RB, Williams DN, Kind AC. Prophylactic antibiotics in hip fractures. A double-blind, prospective study. J Bone Joint Surg Am, 1980; 62(3):457–62. CrossRef
Centers for Disease Control and Prevention (CDC). Surgical site infection event (SSI). 2022. Available via https://www.cdc.gov/nhsn/pdfs/validation/2017/pcsmanual_2017.pdf (Accessed 22 March 2022)
Crawford CB, Clay JA, Seydel AS, Wernberg JA. Surgical site infections in breast surgery: the use of preoperative antibiotics for elective, nonreconstructive procedures. Int J Breast Cancer, 2016; 2016:1645192; doi:10.1155/2016/1645192 CrossRef
Durando P, Bassetti M, Orengo G, Crimi P, Battistini A, Bellina D, Talamini A, Tiberio G, Alicino C, Iudici R, Sticchi C, Ansaldi F, Rossi A, Rosso R, Viscoli C, Icardi G. Adherence to international and national recommendations for the prevention of surgical site infections in Italy: results from an observational prospective study in elective surgery. Am J Infect Control, 2012; 40(10):969–72. CrossRef
Dyar OJ, Huttner B, Schouten J, Pulcini C, ESGAP (ESCMID Study Group for Antimicrobial Stewardship). What is antimicrobial stewardship? Clin Microbiol Infect, 2017; 23(11):793–8. CrossRef
Founou RC, Founou LL, Essack SY. Clinical and economic impact of antibiotic resistance in developing countries: a systematic review and meta-analysis. PLoS One, 2017; 12(12):e0189621; doi: 10.1371/journal.pone.0189621. CrossRef
Goede WJ, Lovely JK, Thompson RL, Cima RR. Assessment of prophylactic antibiotic use in patients with surgical site infections. Hosp Pharm, 2013; 48(7):560–7. CrossRef
Harbarth S, Samore MH. Antimicrobial resistance determinants and future control. Emerg Infect Dis, 2005; 11(6):794–801. CrossRef
Haque M, Sartelli M, McKimm J, Abu Bakar M. Health care-associated infections - an overview. Infect Drug Resist, 2018; 11:2321–33. CrossRef
Herawati F, Yulia R, Hak E, Hartono AH, Michiels T, Woerdenbag HJ, Avanti C. A retrospective surveillance of the antibiotics prophylactic use of surgical procedures in private hospitals in Indonesia. Hosp Pharm, 2019; 54(5):323–9. CrossRef
Hohmann C, Eickhoff C, Radziwill R, Schulz M. Adherence to guidelines for antibiotic prophylaxis in surgery patients in German hospitals: a multicentre evaluation involving pharmacy interns. Infection, 2012; 40(2):131–7. CrossRef
Kanter G, Connelly NR, Fitzgerald J. A system and process redesign to improve perioperative antibiotic administration. Anesth Analg, 2006; 103(6):1517–21. CrossRef
Kaya S, Aktas S, Senbayrak S, Tekin R, Oztoprak N, Aksoy F, Firat P, Yenice S, Oncul A, Gunduz A, Solak S, Kadanali A, Cakar SE, Caglayan D, Yilmaz H, Bozkurt I, Elmaslar T, Tartar AS, Aynioglu A, Kocyigit NF, Koksal I. An evaluation of surgical prophylaxis procedures in Turkey: a multi-center point prevalence study. Eurasian J Med, 2016; 48(1):24–8. CrossRef
Malavaud S, Bonnet E, Vigouroux D, Mounet J, Suc B. L’antibioprophylaxie en chirurgie digestive: audit de pratiques [Prophylactic antibiotic use in gastro-intestinal surgery: an audit of current practice]. J Chir (Paris), 2008; 145(6):579–84. CrossRef
McDonnell Norms Group. Antibiotic overuse: the influence of social norms. J Am Coll Surg, 2008; 207(2):265–75. CrossRef
Musmar SM, Ba’ba H, Owais A. Adherence to guidelines of antibiotic prophylactic use in surgery: a prospective cohort study in North West Bank, Palestine. BMC Surg, 2014; 14:69; doi:10.1186/1471-2482-14-69 CrossRef
Napolitano F, Izzo MT, Di Giuseppe G, Angelillo IF, Collaborative Working Group. Evaluation of the appropriate perioperative antibiotic prophylaxis in Italy. PLoS One, 2013; 8(11):e79532. CrossRef
O’Neill J. Review on AMR. Antimicrobial resistance: tackling a crisis for the health and wealth of nations. 2014. Available via https://amr-review.org/sites/default/files/AMR%20Review%20Paper%20-%20Tackling%20a%20crisis%20for%20the%20health%20and%20wealth%20of%20nations_1.pdf (Accessed 22 March 2022)
Pittalis S, Ferraro F, Piselli P, Ruscitti LE, Grilli E, Lanini S, Ippolito G, Puro V. Appropriateness of surgical antimicrobial prophylaxis in the Latium region of Italy, 2008: a multicenter study. Surg Infect (Larchmt), 2013; 14(4):381–4. CrossRef
Resman F. Antimicrobial stewardship programs; a two-part narrative review of step-wise design and issues of controversy Part I: step-wise design of an antimicrobial stewardship program. Ther Adv Infect Dis, 2020; 7:2049936120933187; doi:10.1177/2049936120933187 CrossRef
Solomon SL, Oliver KB. Antibiotic resistance threats in the United States: stepping back from the brink. Am Fam Physician, 2014; 89(12):938–41.
Telfah S, Nazer L, Dirani M, Daoud F. Improvement in adherence to surgical antimicrobial prophylaxis guidelines after implementation of a multidisciplinary quality improvement project. Sultan Qaboos Univ Med J, 2015; 15(4):e523–7. CrossRef
Tolba YYA, El-Kabbani AO, Al-Kayyali NS. An observational study of perioperative antibiotic-prophylaxis use at a major quaternary care and referral hospital in Saudi Arabia. Saudi J Anaesth, 2018; 12(1):82–8. CrossRef
Tourmousoglou CE, Yiannakopoulou ECh, Kalapothaki V, Bramis J, St Papadopoulos J. Adherence to guidelines for antibiotic prophylaxis in general surgery: a critical appraisal. J Antimicrob Chemother, 2008; 61(1):214–8. CrossRef
Ventola CL. The antibiotic resistance crisis: part 1: causes and threats. P T, 2015; 40(4):277–83.
Wright JD, Hassan K, Ananth CV, Herzog TJ, Lewin SN, Burke WM, Lu YS, Neugut AI, Hershman DL. Use of guideline-based antibiotic prophylaxis in women undergoing gynecologic surgery. Obstet Gynecol, 2013; 122(6):1145–53. CrossRef
Zhang HX, Li X, Huo HQ, Liang P, Zhang JP, Ge WH. Pharmacist interventions for prophylactic antibiotic use in urological inpatients undergoing clean or clean-contaminated operations in a Chinese hospital. PLoS One, 2014; 9(2):e88971. CrossRef
Zhou L, Ma J, Gao J, Chen S, Bao J. Optimizing prophylactic antibiotic practice for cardiothoracic surgery by pharmacists’ effects. Medicine (Baltimore), 2016; 95(9):e2753. CrossRef