INTRODUCTION
Candida species are commensal microorganisms in the human body. However, some predisposing factors could lead to Candida albicans infection called candidiasis. Not only superficial infection but also candidiasis caused by C. albicans is able to cause systemic infection, which contributes to its mortality rate that ranges from 15% to 35% for adults and from 10% to 15% for neonates (Guinea, 2014).
Fluconazole is the first generation of azole drugs which is widely used for candidiasis treatment because it is affordable and has low toxicity (Murray et al., 2015; Paramythiotou et al., 2014). Fluconazole inhibits the biosynthesis of ergosterol by interfering with lanosterol 14-α-demethylase activity to produce toxic 14-α-methyl-3,6-diol (Prasad et al., 2016). However, C. albicans resistance has been reported against fluconazole in recent years (Arora et al., 2017). To maintain its existence, C. albicans overexpress the ERG11 gene, which will upregulate the targeted enzyme and stimulate the efflux pump. Thus, increasing fluconazole concentration is needed to inhibit C. albicans growth. Other than ERG11 gene overexpression, biofilm also plays an important role in C. albicans resistance development and infectivity (Corte et al., 2016). Candida albicans biofilm is an extracellular matrix polymer composed of polysaccharides, amphiphilic chemical components, and other macromolecules (Ramage et al., 2012). The complex structure of this matrix becomes a barrier between C. albicans and the extracellular area. The presence of persistent cells also contributes to C. albicans drug resistance not only against fluconazole but also against other antifungal drugs, such as polyene (Corte et al., 2016).
The emergence of drug resistance necessitates the urgent need for alternative treatment which can inhibit different stages of C. albicans. It has been reported that essential oil showed promising activity as a biofilm inhibitor, such as the essential oils of Cymbopogon citratus, Cedrus sp., and Syzygium aromaticum (Manoharan et al., 2017). Myristica fragrans, an aromatic herbal plant that is traditionally used as a food spice in Southeast Asia, has also been reported to exhibit antimicrobial, antitumor, antioxidant, and antifungal activity (Das et al., 2020; Rodianawati et al., 2015; Salehi et al., 2017). Furthermore, Thileepan et al. (2018) reported that M. fragrans oil exhibited anticandidal activity; however, its inhibition mechanism and combination effect with azoles have not been reported. In this study, we evaluated the antifungal activity of M. fragrans oil against different Candida spp., its inhibition in different biofilm development stages, and the effect of the combination with fluconazole and identified its chemical compounds. In addition, an 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT) assay was performed for evaluating M. fragrans oil activity against different Candida spp., followed by a quantitative polymerase chain reaction (qPCR) assay for assessing the relative C. albicans cell concentration at biofilm inhibition and combination effect activity.
MATERIALS AND METHODS
Candida spp., M. fragrans oil, and control drug
Candida albicans UICC Y-29, Candida glabrata ATCC 66032, and Candida krusei ATCC 6258 cells were grown and maintained in a 90 mm Petri dish containing potato dextrose agar (Difco, USA) at 28°C. For experimental purposes, Candida spp. from the Petri dish were further transferred into slant potato dextrose agar and incubated at a 28°C for 24 hours. Fluconazole (Sigma-Aldrich, Germany) was dissolved in 1% dimethyl sulfoxide (DMSO) (Sibelco, Germany) prior to use. Myristica fragrans oil with a certificate of analysis was purchased from Costumessentialoil, Jakarta, Indonesia. Myristica fragrans oil was diluted in Tween 80 (Sigma-Aldrich®, Germany) to a final tested concentration of 0.2% prior to use.
A 50% inhibitory concentration (IC50) was achieved using a 96-well culture plate coated with 50% fetal bovine serum (FBS) (Sigma-Aldrich, USA). A volume of 100 μl/well of twofold dilution of fluconazole (0.098–50 μg/ml) or M. fragrans oil (0.009%–5% v/v) diluted in Sabouraud dextrose broth (SDB) (Millipore, Germany) was added to a 96-well culture plate, followed by the addition of 100 μl/well of C. albicans (3 × 104 cells) in SDB. The mixture was then incubated at 37°C for 48 hours. The supernatant was carefully aspirated, and then 100 μl of phosphate-buffered saline (PBS) containing 5 μg/μl MTT (Sigma, USA) was added and mixed. After 3 hours of incubation, 100 μl of DMSO was added and the absorbance was measured at 570 nm using a microplate reader (VersaMax, USA).
Inhibition phase activity was evaluated using 96-well culture plates coated with 5% FBS. Candida albicans cell viability was measured using a tetrazolium-based MTT assay, as performed in our previous work (Rahmasari et al., 2020). The evaluation for inhibition at different stages was performed as mentioned below.
Adhesion stage (0–2 hours)
100 μl/well of fluconazole or M. fragrans oil and 100 μl/well of C. albicans (1 × 107 cells) in SDB were added to a 96-well plate coated with 50% FBS. The plate was incubated for 2 hours at 37°C, followed by cell viability measurement by the MTT assay.
Intermediate stage (2–18 hours)
100 μl/well of C. albicans (1 × 107 cells) in SDB was added to a 96-well plate coated with 50% FBS. The plate was incubated for 2 hours at 37°C. The supernatant was then aspirated, followed by the addition of fresh SDB and 100 μl/well fluconazole or M. fragrans oil and 100 μl/well SDB. The plate was incubated for 16 hours at 37°C, followed by the MTT assay.
Maturation stage (24–48 hours)
100 μl/well of C. albicans (1 × 107 cells) in SDB was added to a 96-well plate coated with 50% FBS. The plate was incubated for 24 hours at 37°C, followed by the addition of 100 μl/well fluconazole or M. fragrans oil. The plate was further incubated for 24 hours at 37°C, followed by the MTT assay.
MTT assay
After incubation, the supernatant was carefully aspirated, followed by the addition of 100 μl PBS containing 5 μg/μl of MTT, and incubated for 3 hours at room temperature. After incubation, 100 μl DMSO was added and absorbance was measured at 570 nm using a microplate reader.
Drug combination
These effects were further determined by the interaction index value, as described by Tallarida (2002) and as performed in our previous work (Makau et al., 2018). In order to calculate the interaction index for the combination of drugs A and B, the following equation was used: Ac/Ae + Bc/Be = γ, where Ac and Bc correspond to the concentrations of A and B when used in combination; Ae and Be correspond to the concentrations able to produce an effect of the same magnitude if used alone; and γ corresponds to the interaction index value. If γ is <1, the effect of the combination is synergistic, whereas if γ = or >1, the effect is additive or antagonistic, respectively. The evaluation was carried out using a 96-well culture plate coated with 50% FBS. In brief, 100 μl/well of 3 × 104 C. albicans cells in SDB was added to 100 μl/well of a mixture containing twofold dilution of fluconazole (0.098–50 μg/ml) and M. fragrans oil (0.047%–3% v/v) diluted in SDB. The mixture was then incubated at 37°C for 48 hours. The supernatant was aspirated, and then 50 μl of MTT was added and mixed. After 3 hours of incubation, 100 μl of DMSO was added and the absorbance was measured at 570 nm using a microplate reader.
Quantitative real time polymerase chain reaction
Candida albicans cultures from the evaluation on 96-well plates were collected into microcentrifuge tubes and centrifuged (Sorvall® Fresco, USA) at 4,000 rpm for 10 minutes. The supernatants were discarded and the pellets that formed were extracted using the YeaStar Genomic DNA KitTM (Zymo Research, USA). The extracted DNAs were used as DNA templates. Two pairs of specific primers from the internally transcribed spacer (ITS) SACALF (ITS-1) 5′-TTTATCAACTTGTCACACCAGA-3′ and SACALR (ITS-2) 5′-GGTCAAAGTTTGAAGATATACGT-3′ were used based on research conducted by Asadzadeh et al. (2018). The DNA samples were then amplified using THUNDERBIRDTM SYBRTM qPCR Mix (Toyobo, Japan) with the MA-600 Real-Time Quantitative Thermal Cycler (Molarray Biotech, China). The qPCR cycle settings were predenaturation at 95°C for 30 seconds, followed by 40 cycles of denaturation at 95°C for 15 seconds, annealing at 55°C for 30 seconds, and extension at 72°C for 30 seconds. All samples were run in duplicate. A standard curve was made using standard solutions from serial dilutions (102–106 CFU/ml) to obtain a linear regression equation from plotting the cycle threshold (Ct) value against a predetermined CFU value (cell concentration). The results were presented as relative cell concentration, which was determined as (CFU value of treated sample divided by CFU value of untreated sample) × 100%.
Chemical analysis of M. fragrans oil
This was performed by gas chromatography-mass spectroscopy (GC-MS) using TRACE GC Ultra with flame ionization detector (FID), MS DSQ II detector, and MS-FID splitter with an Rtx-1 MS column (Restek, 60 m × 0.25 mm ID, film thickness 0.25 μm) using helium as a carrier gas and an electron impact ionization mode at 70 eV. The temperature of the injector was kept at 280°C, ion source temperature at 200°C, FID temperature at 300°C, and temperature programming at 2°C/minute from 50°C to 310°C. The identification of the compounds was based on the comparison of their retention times and mass spectra with those stored in the GC/MS data system and National Institute of Standards and Technology. The compound percentages were computed from the total area of the peak by a GC/MS software apparatus.
Statistical analysis
Data are represented as the mean ± standard error from two independent experiments (each in duplicate). Data were analyzed using GraphPad Prism 9.1.0.
RESULTS
M. fragrans activity against C. albicans
We used fluconazole as a positive control and determined the inhibitory activity of M. fragrans oil against various Candida species using the MTT assay. The comparison of treated C. albicans viability to untreated C. albicans was analyzed using GraphPad Prism 9.1.0. The results showed that the IC50 of fluconazole was 5.05 ± 2.22 μg/ml and M. fragrans oil showed an IC50 of 1.76% ± 0.44% (v/v) against C. albicans, as shown in Table 1, respectively. In order to check the spectrum activities of fluconazole and M. fragrans oil against Candida spp., their inhibitory activity was tested against C. glabrata and C. krusei. All Candida spp. tested were sensitive to fluconazole, while M. fragrans oil was more effective against C. albicans and C. krusei than C. glabrata (Table 1).
Furthermore, the inhibitory activity was also evaluated by quantifying the relative cell concentration of C. albicans using the qPCR method. The cell concentration was obtained from the standard curve (Supplementary Data). The inhibitory activity was evaluated by comparing the cell concentration of the treated C. albicans to that of the untreated C. albicans (Liu et al., 2011). Candida albicans was treated with fluconazole at concentrations of 12.5, 0.4, and 0 μg/ml and M. fragrans oil at concentrations of 2.5%, 0.6%, and 0% v/v. Based on the qPCR results, it was found that fluconazole with concentrations of 0.4 and 12.5 μg/ml inhibited the growth of C. albicans up to 40.6% and 72.2% with percentages of relative cell concentration at 59.4% ± 6.6% and 27.8% ± 2.4%, as shown in Figure 1A. Meanwhile, in M. fragrans oil, concentrations of 0.6% and 2.5% v/v could inhibit the growth of C. albicans up to 23.9% and 67.8% with percentages of relative cell concentration at 76.1% ± 1.1% and 32.2% ± 4.2%, as shown in Figure 1B, respectively.
Inhibitory activity of M. fragrans oil against C. albicans biofilm formation
The inhibitory activity of fluconazole and M. fragrans oil against C. albicans biofilm formation is shown in Figure 2. Analysis using the MTT assay showed that the addition of fluconazole 12.56 μg/ml decreased the viability of C. albicans to 86.77% at the cell adhesion, 79.36% at intermediate, and 76.59% at maturation phase compared to the untreated cells. Meanwhile, the addition of 3.1% (v/v) M. fragrans oil decreased the viability of C. albicans to 13.08% at the cell adhesion phase and to 19.84% at the intermediate phase and did not inhibit the maturation phase compared to the untreated cells (Fig. 2A).
The qPCR quantification showed that fluconazole (Fig. 2B) mainly showed an inhibition effect at the C. albicans biofilm maturation stage with relative cell concentration decrease to 39.7% ± 0.4% compared to the untreated C. albicans. Meanwhile, M. fragrans oil (Fig. 2B) exhibited the pronounced activity at the adhesion and intermediate stages with relative cell concentrations at 39.2% ± 12.4% and 11.8% ± 3.6% compared to the untreated cells. Mild activity was observed at the maturation phase (76% ± 17.4% relative cell concentration). The MTT assay and cell quantification by qPCR suggest that M. fragrans oil targets the adhesion and biofilm intermediate stages.
Combination effect of M. fragrans oil and fluconazole against C. albicans
Myristica fragrans oil and fluconazole exhibited activity towards different stages of biofilm maturation. We therefore performed combination studies to determine the effect of M. fragrans oil and fluconazole cotreatment on C. albicans. As shown in Table 2, the addition of M. fragrans oil potentiates the inhibitory effect of fluconazole, as determined by the MTT assay. The IC50 of fluconazole and M. fragrans oil alone is 5.05 μg/ml and 1.76% (v/v), respectively. Therefore, the isobole method was used to calculate the interaction index at 50% inhibitory activity to determine whether the cotreatment resulted in a synergistic effect or not. The addition of 0.047% of M. fragrans oil could reduce the IC50 of fluconazole against C. albicans by more than half to 2.82 μg/ml, resulting in an interaction index of 0.49, which is synergistic. As indicated in Table 2, other combinations also showed an interaction index of <1, denoting a synergistic effect.
 | Table 1. Activity of M. fragrans oil against Candida spp. [Click here to view] |
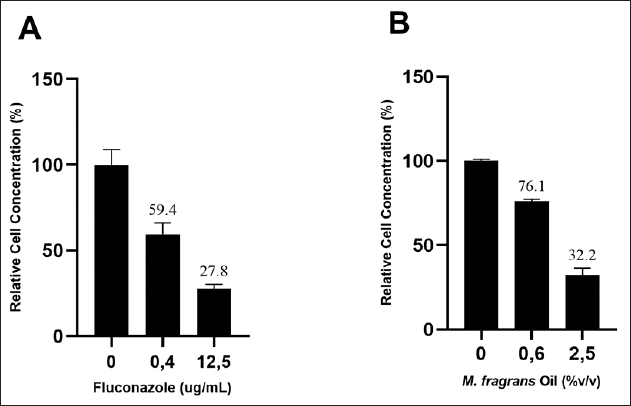 | Figure 1. Relative C. albicans cell concentration (quantified by qPCR) after treatment with fluconazole and M. fragrans oil incubated in 37°C incubator for 48 hours. The percentages of cell concentration after treatment with (A) fluconazole and (B) M. fragrans oil were obtained by comparing the treated to untreated C. albicans. Data represented as mean ± SD from two independent experiments, each done in duplicate. [Click here to view] |
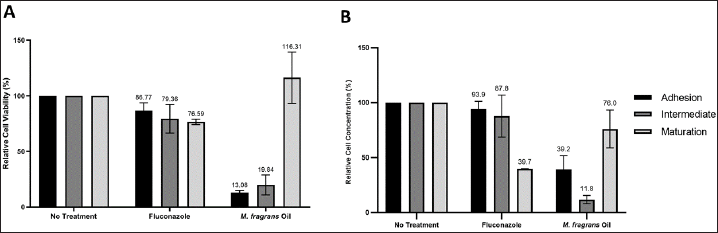 | Figure 2. Inhibitory activity of 12.56 μg/ml fluconazole and 3.1% (v/v) M. fragrans oil against C. albicans biofilm formation at adhesion, intermediate, and maturation stages as evaluated by (A) MTT assay and (B) qPCR. Adhesion indicated by black color, intermediate indicated by dark-gray color, and maturation phase indicated by light-gray color. Data represented as mean ± SD from two independent experiments, each in duplicate. [Click here to view] |
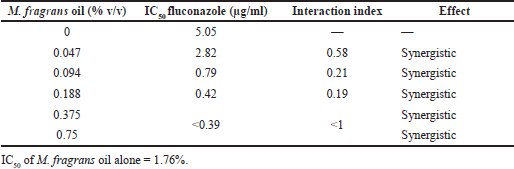 | Table 2. Combination effect of fluconazole with M. fragrans oil. [Click here to view] |
In addition, we used qPCR to determine the relative concentration of the C. albicans cells in a combined treatment of 0.39 μg/ml fluconazole and 0.8%, 0.4%, and 0.2% (v/v) of M. fragrans oil. The IC50 of fluconazole is 5.05 μg/ml. Therefore, we used 0.39 μg/ml, a much lower concentration, to show the synergistic effect of M. fragrans oil. As shown in Figure 3, the addition of various concentrations of M. fragrans oil greatly reduced the cell concentration of C. albicans, which could be detected by qPCR.
Chemical analysis of M. fragrans oil
Identification of potential active components in M. fragrans oil was performed by gas chromatography. Based on the GC-MS analysis, shown in Table 3, respectively, myristicin (11.81%) and α-copaene (11.47%) existed as major compounds, followed by caryophyllene (6.84%), α-pinene (6.15%), and sabinene (5.98%).
DISCUSSION
The incidence of candidiasis has increased in the last few decades (de Oliveira Santos et al., 2018). Since late 1990, the emergence of resistance among Candida species has been reported, especially against azole drugs (Orozco et al., 1998). Candida albicans, the main cause of candidiasis, is able to adapt to environmental changes by polymorphism, proteolytic enzyme production, phenotype switch, and biofilm formation (Clark-Ordóñez et al., 2017). It was reported that some essential oils, as mentioned above, show potential inhibitory effects against Candida spp. biofilm by inhibiting hyphal formation, altering genes related to biofilm formation, or inhibiting adhesive proteins (Arora et al., 2017; Clark-Ordóñez et al., 2017; El-Baz et al., 2021). In the present research, we used relative cell viability and cell quantification by the MTT assay and qPCR, respectively, to demonstrate that M. fragrans oil has potent inhibition activity against C. albicans, which is in line with a previous report (Thileepan et al., 2017). In addition, we showed that M. fragrans oil exhibits activity against other Candida species, C. krusei and C. glabrata (Table 1), indicating its broad spectrum of activity. The IC50 of fluconazole against C. krusei was higher than those of C. albicans and C. glabrata. It has previously been reported that the activity of 14α-demethylase in C. krusei is more resistant to inhibition by fluconazole than in C. albicans strains (Orozco et al., 1998). However, M. fragrans oil showed potent activity against C. albicans and C. krusei more than against C. glabrata. Candida albicans and C. krusei were found to have the ability to produce extracellular DNase (DNA cutting enzyme), but no such ability in C. glabrata (Riceto et al., 2015). Extracellular DNA in Candida spp. is known to have an important role in the integrity and maintenance of the biofilm structure (Martins et al., 2010; Sapaar et al., 2014). Therefore, the sensitivity of C. albicans and C. krusei to M. fragrans oil compared to C. glabrata may be due to its components that affect the extracellular DNase activity so that it disrupts biofilm integrity, but further research is needed to prove this.
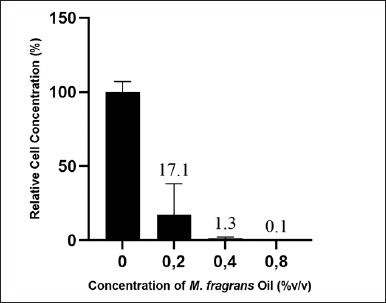 | Figure 3. Relative C. albicans cell concentration (quantified by qPCR) after treatment with 0.39 μg/ml fluconazole and various concentrations of M. fragrans oil. Data represented as mean ± SD from two independent experiments. [Click here to view] |
Candida albicans biofilm is formed in three phases, which are the adhesion, intermediate, and maturation phases. In the adhesion phase, C. albicans cells adhere to the surface of the host during the first 1-2 hours. The intermediate or biofilm development phase occurs for up to approximately 24 hours, followed by the maturation phase in which biofilms with complex structures and compositions exist. The maturation phase occurs between 24 and 72 hours after the attachment of the C. albicans cells (Cavalheiro and Teixeira, 2018; Mayer et al., 2013). In the current research, fluconazole, as a control drug, mainly inhibited the intermediate and maturation phases of C. albicans biofilm more than the adhesion phase (Fig. 2A), as confirmed by qPCR (Fig. 2B). According to research by Khodavandi et al. (2011), a significant decrease in C. albicans cells treated with fluconazole was observed only after 4 hours incubation compared to the untreated controls, which is in accordance with the results of experiments carried out previously. This phenomenon is related to the mechanism of the azole class to inhibit lanosterol demethylase (14-α-sterol demethylase). When this enzyme is inhibited, other enzymes in the ergosterol biosynthetic pathway will synthesize toxic sterols, namely, 14α-methylergosta-8-24-(28)-dinol, which is fungistatic; therefore, it takes a longer time for fluconazole to have the effect of reducing fungal cell viability (Bhattacharya et al., 2020; Ellepola et al., 2015; Lu et al., 2021). On the other hand, M. fragrans oil was confirmed to mainly inhibit the adhesion and intermediate phases (Fig. 2A and B). In addition, the inhibitory activity of fluconazole and M. fragrans oil against C. albicans biofilm formation at the adhesion to intermediate phases (0–18 hours) was also evaluated. The continuous incubation of fluconazole or M. fragrans oil showed better activity than when the drug or oil was added at each stage only (Supplementary Data).
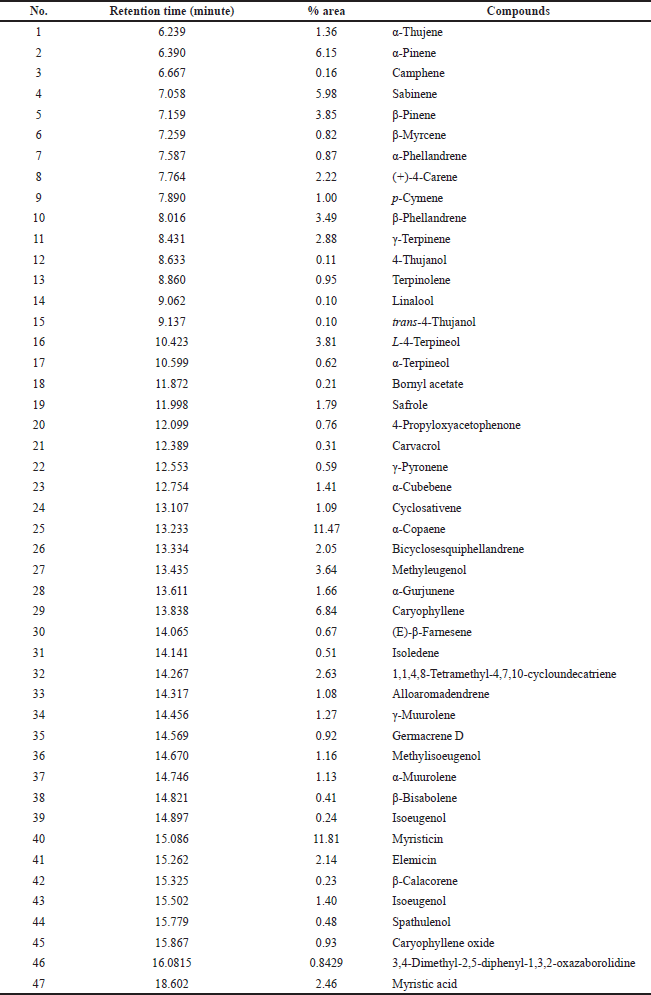 | Table 3. Phytochemical compositions of M. fragrans oil by GC/MS analysis. [Click here to view] |
GC-MS demonstrated the presence of sesquiterpenes, especially α-copaene (11.47%) and caryophyllene (6.84%), as shown in Table 3, respectively. Those M. fragrans oil sesquiterpenes may contribute to adhesion phase inhibition of C. albicans, as the sesquiterpenes from Carpesium macrocephalum have been reported to inhibit the morphogenetic transformation of yeast to hyphae form (Xie et al., 2015). In addition, M. fragrans oil is known to contain α-pinene, β-pinene, sabinene, and myristicin (Table 3), which possibly inhibited the adhesion and intermediate phases (de Macêdo Andrade et al., 2018; Hammer et al., 2003; Moreira Valente et al., 2015; Thakre et al., 2018). α-Pinene compounds were found to inhibit DNA replication, RNA synthesis, polysaccharides in cell walls, and ergosterol in the cytoplasmic membrane and have inhibitory activity on biofilm formation of C. albicans. The compound (+)-β-pinene most likely acts by disrupting cell walls through molecular interactions with delta-14-sterol reductase and, to a lesser extent, with 1,3-β-glucan synthase (de Macêdo Andrade et al., 2018). In addition, myristicin and sabinene, which are the main compounds in M. fragrans oil, are fungicidal with unknown mechanisms (Moreira Valente et al., 2015; Zhou et al., 2019). However, M. fragrans oil did not exhibit activity at the maturation phase of C. albicans, possibly because the biofilm matrix had already formed, thus blocking or slowing the rate of the diffusion of the external factors (Limoli et al., 2015; Pinto et al., 2020). In addition, hyphal cells in C. albicans biofilms express antioxidant defense proteins (e.g., superoxide dismutase 5) that play a role in the detoxification of reactive oxygen species. These proteins are able to block off limonene activity to produce oxidative stress-induced cell apoptosis (Noble et al., 2017; Thakre et al., 2018).
The combination of antifungal therapies, which has different targets and works synergistically, is one of the strategies to overcome drug resistance (Pai et al., 2018). The combination of fluconazole at lower concentrations with M. fragrans oil resulted in a synergistic effect (Table 2). Quantification of the C. albicans genetic material in a combined treatment of M. fragrans oil and fluconazole (Fig. 3) showed a strong synergistic effect, which could result from different mechanisms of action of both drugs (Pai et al., 2018). In Figure 2B, M. fragrans oil exhibited a strong inhibitory effect on the adhesion and intermediate phases; meanwhile, fluconazole exhibited an inhibitory effect mainly at the maturation phase. Therefore, a combination of both could produce a strong synergy against C. albicans. The concomitant use of fluconazole and M. fragrans oil for candidiasis treatment can reduce C. albicans resistance against fluconazole due to their different mechanism of action. Further, M. fragrans oil can be used topically as an adjunctive therapy in mucocutaneous and oropharyngeal candidiasis treated with oral fluconazole.
CONCLUSION
The result presented here demonstrated clearly that M. fragrans oil possesses potent activity against C. albicans by inhibiting biofilm formation and could be used as an antifungal agent in combination with fluconazole, as emphasized by its synergistic effect. Further research on its potential active compound activity against C. albicans infection, safety, and effectiveness in vivo is needed.
ACKNOWLEDGMENTS
This research was partially funded by the PUTI Saintekes Grant 2020 from Universitas Indonesia.
CONFLICTS OF INTEREST
The authors declare that there are no conflicts of interest.
ETHICAL APPROVAL
No informed consent or animal ethical statement was needed for this research.
DISCLOSURE
All authors certify that they have no financial or proprietary interests in any material discussed in this article.
AUTHOR CONTRIBUTIONS
All authors made substantial contributions to conception and design, acquisition of data, or analysis and interpretation of data; took part in drafting the article or revising it critically for important intellectual content; agreed to submit to the current journal; gave final approval of the version to be published; and agree to be accountable for all aspects of the work. All the authors are eligible to be an author as per the international committee of medical journal editors (ICMJE) requirements/guidelines.
DATA AVAILABILITY
All data generated and analyzed are included within this research article.
PUBLISHER’S NOTE
This journal remains neutral with regard to jurisdictional claims in published institutional affiliation.
REFERENCES
Arora S, Dhuria N, Jindal N, Galhotra S. Speciation, biofilm formation and antifungal susceptibility of Candida isolates. Int J Res Dev Pharm Life Sci, 2017; 6:2; doi:10.21276/IJRDPL.2278-0238.2017.6(2).2517-2521 CrossRef
Asadzadeh M, Ahmad S, Al-Sweih N, Khan Z. Rapid and accurate identification of Candida albicans and Candida dubliniensis by real-time PCR and melting curve analysis. Med Princ Pract, 2018; 27:543–8; doi:10.1159/000493426 CrossRef
Bhattacharya S, Sae-Tia S, Fries BC. Candidiasis and mechanisms of antifungal resistance. Antibiotics (Basel), 2020; 9:312; doi:10.3390/antibiotics9060312 CrossRef
Cavalheiro M, Teixeira MC. Candida biofilms: threats, challenges, and promising strategies. Front Med (Lausanne), 2018; 5:28; doi:10.3389/fmed.2018.00028 CrossRef
Clark-Ordóñez I, Callejas-Negrete OA, Aréchiga-Carvajal ET, Mouriño-Pérez RR. Candida species diversity and antifungal susceptibility patterns in oral samples of HIV/AIDS patients in Baja California, Mexico. Med Mycol, 2017; 55:285–94; doi:10.1093/mmy/myw069 CrossRef
Corte L, Roscini L, Colabella C, Tascini C, Leonildi A, Sozio E, Menichetti F, Merelli M, Scarparo C, Meyer W. Exploring ecological modelling to investigate factors governing the colonization success in nosocomial environment of Candida albicans and other pathogenic yeasts. Sci Rep, 2016; 6:26860; doi: 10.1038/srep26860 CrossRef
de Macêdo Andrade AC, Rosalen PL, Freires IA, Scotti L, Scotti MT, Aquino SG, de Castro RD. Antifungal activity, mode of action, docking prediction and anti-biofilm effects of (+)-β-pinene enantiomers against Candida spp. Curr Top Med Chem, 2018; 18:2481–90; doi:10.2174/1568026618666181115103104 CrossRef
de Oliveira Santos GC, Vasconcelos CC, Lopes AJO, de Sousa Cartágenes MDS, Filho AKDB, do Nascimento FRF, Ramos RM, Pires ERRB, de Andrade MS, Rocha FMG, de Andrade Monteiro C. Candida infections and therapeutic strategies: mechanisms of action for traditional and alternative agents. Front Microbiol, 2018; 9:1351; doi:10.3389/fmicb.2018.01351 CrossRef
El-Baz AM, Mosbah RA, Goda RM, Mansour B, Sultana T, Dahms TES, El-Ganiny AM. Back to nature: combating Candida albicans biofilm, phospholipase and hemolysin using plant essential oils. Antibiotics (Basel), 2021; 10:81; doi:10.3390/antibiotics10010081 CrossRef
Ellepola AN, Khajah R, Jayatilake S, Samaranayake L, Sharma P, Khan Z. Impact of brief exposure to antifungal agents on the post-antifungal effect and hemolysin activity of oral Candida albicans. J Appl Oral Sci, 2015; 23:412–8; doi:10.1590/1678-775720150100 CrossRef
Guinea J. Global trends in the distribution of Candida species causing candidemia. Clin Microbiol Infect, 2014; 20(Suppl. 6):5–10; doi: 10.1111/1469-0691.12539 CrossRef
Hammer KA, Carson CF, Riley TV. Antifungal activity of the components of Melaleuca alternifolia (tea tree) oil. J Appl Microbiol, 2003; 95:853–60; doi:10.1046/j.1365-2672.2003.02059.x CrossRef
Khodavandi A, Alizadeh F, Harmal NS, Sidik SM, Othman F, Sekawi Z, Jahromi MAF, Ng KP, Chong PP. Comparison between efficacy of allicin and fluconazole against Candida albicans in vitro and in a systemic candidiasis mouse model. FEMS Microbiol Lett, 2011; 315:87–93; doi:10.1111/j.1574-6968.2010.02170.x CrossRef
Limoli DH, Jones CJ, Wozniak DJ. Bacterial extracellular polysaccharides in biofilm formation and function. Microbiol Spectr, 2015; 3; doi:10.1128/microbiolspec.MB-0011-2014 CrossRef
Liu G, Xiong S, Xiang YF, Guo CW, Ge F, Yang CR, Zhang YJ, Wang YF, Kitazato K. Antiviral activity and possible mechanisms of action of pentagalloylglucose (PGG) against influenza A virus. Arch Virol, 2011; 156:1359–69; doi:10.1007/s00705-011-0989-9 CrossRef
Lu H, Shrivastava M, Whiteway M, Jiang Y. Candida albicans targets that potentially synergize with fluconazole. Crit Rev Microbiol, 2021; 47:323–37; doi:10.1080/1040841X.2021.1884641 CrossRef
Makau JN, Watanabe K, Mohammed MMD, Nishida N. Antiviral activity of peanut (Arachis hypogaea L.) skin extract against human influenza viruses. J Med Food, 2018; 21:777–84; doi:10.1089/jmf.2017.4121 CrossRef
Manoharan RK, Lee JH, Kim YG, Kim SI, Lee J. Inhibitory effects of the essential oils α-longipinene and linalool on biofilm formation and hyphal growth of Candida albicans. Biofouling, 2017; 33:2, 143–55; doi:10.1080/08927014.2017.1280731 CrossRef
Martins M, Uppuluri P, Thomas DP, Cleary IA, Henriques M, Lopez-Ribot JL, Oliveira R. Presence of extracellular DNA in the Candida albicans biofilm matrix and its contribution to biofilms. Mycopathologia, 2010; 169:323–31; doi:10.1007/s11046-009-9264-y CrossRef
Mayer FL, Wilson D, Hube B. Candida albicans pathogenicity mechanisms. Virulence, 2013; 4:119–28; doi:10.4161/viru.22913 CrossRef
Moreira Valente VM, Jham GN, Jardim CM, Dhingra O, Ghiviriga I. Major antifungals in nutmeg essential oil against Aspergillus flavus and A. ochraceus. J Food Res, 2015; 4:51–7; doi:10.5539/jfr.v4n1p51 CrossRef
Murray P, Rosenthal K, Pfaller M. Medical microbiology. 8th edition, Elsevier, Philadelphia, PA, 2015.
Noble SM, Gianetti BA, Witchley JN. Candida albicans cell-type switching and functional plasticity in the mammalian host. Nat Rev Microbiol, 2017; 15:96–108; doi:10.1038/nrmicro.2016.157 CrossRef
Orozco AS, Higginbotham LM, Hitchcock CA, Parkinson T, Falconer D, Ibrahim AS, Ghannoum MA, Filler SG. Mechanism of fluconazole resistance in Candida krusei. Antimicrob Agents Chemother, 1998; 42:2645–9; doi:10.1128/AAC.42.10.2645 CrossRef
Pai V, Ganavalli A, Kikkeri NN. Antifungal resistance in dermatology. Indian J Dermatol, 2018; 63:361–8; doi:10.4103/ijd.IJD_131_17 CrossRef
Paramythiotou E, Frantzeskaki F, Flevari A, Armaganidis A, Dimopoulos G. Invasive fungal infections in the ICU: how to approach, how to treat. Molecules, 2014; 19:1085–119; doi:10.3390/molecules19011085 CrossRef
Pinto RM, Soares FA, Reis S, Nunes C, Van Dijck P. Innovative strategies toward the disassembly of the EPS matrix in bacterial biofilms. Front Microbiol, 2020; 11:952; doi:10.3389/fmicb.2020.00952 CrossRef
Prasad R, Shah AH, Rawal MK. Antifungals: mechanism of action and drug resistance. Adv Exp Med Biol, 2016; 892:327–49; doi:10.1007/978-3-319-25304-6_14 CrossRef
Rahmasari R, Chairunissa AH, Irianti MI, Forestrania RC, Arifianti A, Suryadi H, Makau JN, Jantan I, Elya B. Inhibitory and anti-biofilm effects of Orthoshipon aristatus against Candida albicans. Pharm Sci Res, 2020; 7(3):132–7; doi:10.7454/psr.v7i3.1104 CrossRef
Ramage G, Rajendran R, Sherry L, Williams C. Fungal biofilm resistance. Int J Microbiol, 2012; 2012:528521; doi: 10.1155/2012/528521 CrossRef
Rodianawati I, Hastuti P, Cahyanto MN. Nutmeg’s (Myristica fragrans Houtt) oleoresin: effect of heating to chemical compositions and antifungal properties. Proc Food Sci, 2015; 3:244–54. CrossRef
Salehi B, Upadhyay S, Orhan IE, Jugran AK, Jayaweera SLD, Dias DA, Sharopov F, Taheri Y, Martins N, Baghalpour N, Cho WC, Rad JS. Therapeutic potential of α- and β-pinene: a miracle gift of nature. Biomolecules, 2019; 9(11):738; doi:10.3390/biom9110738 CrossRef
Riceto ÉB, Menezes Rde P, Penatti MP, Pedroso Rdos S. Enzymatic and hemolytic activity in different Candida species. Rev Iberoam Micol, 2015; 32:79–82; doi:10.1016/j.riam.2013.11.003 CrossRef
Sapaar B, Nur A, Hirota K, Yumoto H, Murakami K, Amoh T, Matsuo T, Ichikawa T, Miyake Y. Effects of extracellular DNA from Candida albicans and pneumonia-related pathogens on Candida biofilm formation and hyphal transformation. J Appl Microbiol, 2014; 116:1531–42; doi:10.1111/jam.12483 CrossRef
Tallarida RJ. The interaction index: a measure of drug synergism. Pain, 2002; 98:163–8; doi:10.1016/s0304-3959(02)00041-6 CrossRef
Thakre A, Zore G, Kodgire S, Kazi R, Mulange S, Patil R, Shelar A, Santhakumari B, Kulkarni M, Kharat K, Karuppayil SM. Limonene inhibits Candida albicans growth by inducing apoptosis. Med Mycol, 2018; 56:565–78; doi:10.1093/mmy/myx074 CrossRef
Thileepan T, Thevanesam V, Kathirgamanathar S. Antimicrobial activity of seeds and leaves of Myristica fragrans against multi-resistant microorganisms. J Agric Sci Technol A, 2017; 7:302–8; doi: 10.17265/2161-6256/2017.05.002 CrossRef
Xie C, Sun L, Meng L, Wang M, Xu J, Bartlam M, Guo Y. Sesquiterpenes from Carpesium macrocephalum inhibit Candida albicans biofilm formation and dimorphism. Bioorg Med Chem Lett, 2015; 25:5409–11; doi:10.1016/j.bmcl.2015.09.013 CrossRef
Zhou S, Wei C, Zhang C, Han C, Kuchkarova N, Shao H. Chemical composition, phytotoxic, antimicrobial and insecticidal activity of the essential oils of Dracocephalum integrifolium. Toxins (Basel), 2019; 11; doi:10.3390/toxins11100598 CrossRef
SUPPLEMENTARY DATA
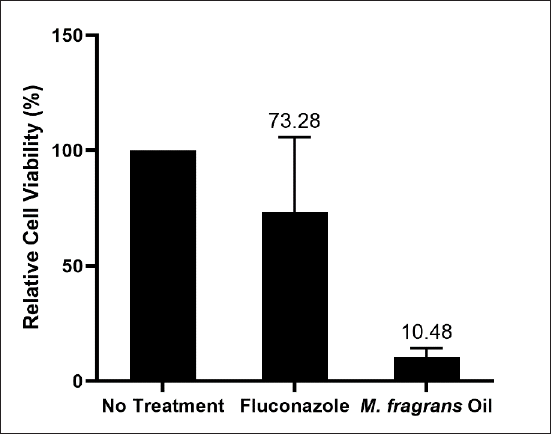 | Supplementary Data. Inhibitory activity of 12.56 μg/ml fluconazole and 3.1% (v/v) M. fragrans oil against C. albicans biofilm formation at the adhesion to intermediate stages (0–18 hours) as evaluated by MTT assay. The experiment was performed as follows: 100 μl/well of fluconazole or M. fragrans oil and 100 μl/well of C. albicans (1 × 107 cells) in SDB were added into a 96-well plate coated with 50% FBS. The plate was incubated for 18 hours at 37°C, followed by cell viability measurement by the MTT assay. Data represented as mean ± SD from two independent experiments, each in duplicate. [Click here to view] |