INTRODUCTION
Parkinson’s disease (PD) is the second most common neurodegenerative disorder characterized by the progressive death of dopaminergic neurons. PD is found in all ethnic groups; however, geographical differences are observed. Early onset of sporadic Alzheimers disease is rare and about 4% patients develop clinical sign before the age of 50 years. About 1%–2% of the age group of 65 years develops PD and this increases to 3%–5% in the age group of 85 years (Alves et al., 2008). Clinical features of PD will appear when striatal dopaminergic levels are largely reduced or lost. Abnormal cellular components like oxidative stress, impaired protein disposal system, and chronic inflammation may be responsible for pathogenesis underlying neurodegeneration (Schintu et al., 2009). Peroxisome Proliferative Activated Receptor-gama (PPAR-γ) present in large variety of cells and maximum level of expression is in adipose tissue, peripheral and central nervous system cells. The regulation of glucose and lipid metabolism by PPAR-γ depends on its distribution patterns (Straus and Glass, 2007). The presence of PPAR-γ in basal ganglia and areas expressing dopamine receptor with high concentration supports the hypothesis of involvement of PPAR-γ in pathogenesis of PD. This increases the interest for PPAR-γ agonists in PD management (Moreno et al., 2004). Neuroinflammation and microglia activation plays an important role in PD pathogenesis (Hirsch and Hunot, 2009), but currently there are no drugs that will directly tackle the inflammatory component of PD. There is a need for the development of new therapeutics which acts via novel target(s) (may be like neuroinflammation) and gives more benefits than the existing drugs. PPAR-γ found to have a prominent role in the biogenesis of mitochondria (Hu and Wang, 2016), and it can be a powerful target in the treatment of many neurodegenerative diseases including PD. It was also reported to have anti-inflammatory and anti-oxidant activity but the mechanism is not yet established (Amin et al., 2017; Corona and Duchen, 2015). Based on the above hypothesis, the present study was conceived to evaluate novel PPAR-γ agonists for their protective effect against rotenone-induced toxicity in mouse model of PD.
MATERIALS AND METHODS
Novel glitazone compounds were obtained as gift samples from Dr. Prashanth Kumar B R, Assistant Professor, Department of Pharmaceutical Chemistry, JSS College of Pharmacy, Mysore. The experiments were carried out on Wister Albino Rats and Balb/c mice. The studies conducted were approved by the Institutional Animal Ethics Committee of JSS College of Pharmacy, Mysore, Karnataka. (Approval no: IAEC/JSSCPM/296/2018).
Molecular docking studies
The docking studies were performed by using the SYBYL X 2.1.1 (Licensed software) software package platform for 14 virtual glitazones (Table 1). The selected glitazones for the present study from docking results are as shown in the Figure 1. The proteins were downloaded from the protein data bank and prepared by using BIOPOLYMER PREPARATION wizard. Proteins were prepared by joining the missing loops and by adding the hydrogen and deleting water molecules; then energy minimization was performed by adding the charges. All these processes were done to bring the protein to its naturally existing state (Naim et al., 2018).
The ligands were prepared by using the LIGAND PREPARATION wizard in which the ligands were converted to their 3D conformation and by minimizing the energy they had bought to the stable form and finally, the charges were added. The docking operation was performed on DOCK LIGAND wizard by using the SYBYL X 2.1.1 software package platform wherein the prepared ligand and proteins were taken for further study. By using the PROTOMAL GENERATION wizard, the active site of the protein was identified and the ligands were docked along with the co-crystallized ligand which was present within the crystal structure of protein molecule. Finally, the binding poses were observed, and the docking score was obtained by using the SYBYL X 2.1.1 scoring function (Amin et al., 2017).
Pharmacokinetic study of selected glitazones
The kinetic study was carried out for the two test compounds (C25 and C34) and one standard pioglitazone (Fujita et al., 2003; Pandit et al., 2012). The study was conducted in Wister rats weighing between 200– and 250 g. All test substances were given as Sod CMC suspension orally at 10 mg/kg. The blood samples were collected in the ethylene diamine tetraacetic acid containing tubes by tail vein method (Rangaraj et al., 2014) at different time intervals of 0, 1, 2, 4, 8, 16, and 24-hour. The blood samples were centrifuged by using the cooling centrifuge and stored in the freezer at −20°C until analysis. The blood samples were analyzed using the high-performance liquid chromatography method.
Acute oral toxicity study
The acute oral toxicity study was done by OECD 423 guideline for newly derived novel glitazone compounds C25 and C34 (OECD guideline 423, 2018).
Anti-Parkinson’s activity of novel glitazone
After acclimatization the animals were weighed and based on the bodyweight of the animals, they were grouped by randomization (n = 6) (Cannon et al., 2009). The study duration was 21 days, and at the end of the study the animals were sacrificed and the brain of the animals was used for estimation of endogenous antioxidant activity. The behavioral activities of the animals were carried out on day 0, day 7, day 14, and day 21 of the study. The study protocol is given in Table 2.
RESULTS AND DISCUSSION
Molecular docking study
The docking studies revealed the binding ability of novel compounds C25 and C34, which showed better interaction with PPARγ receptors than the other compounds when compared with the reference ligand (rosiglitazone). The standard levodopa has shown the less binding activity when compared with reference ligand because of its less binding activity with the target protein 3CS8 (Table 3 and Fig. 2). It was observed from docking that without ligand binding to the legend binding domain (LBD) of PPAR-γ the structure of the adjacent helix, C-terminal helix and PGC-1α does not show any structural variation. On the other hand, any agonist binding to the PPAR-γ LBD, such as compound C25, compound C34, and rosiglitazone shows the transformation of the secondary structure of the protein. Therefore, docking has clearly demonstrated the usefulness of these glitazones in neurodegenerative disorders especially in PD
Pharmacokinetic study
The pharmacokinetic study was conducted to know the pharmacokinetic behavior of the novel glitazones derivatives. The two compounds C25 and C34 were taken based on the best results from molecular docking study, and the kinetic study was done for these compounds. The compound C25 has shown good kinetic profile over C34 (Table 4 and Fig. 3).
Acute toxicity study
Acute toxicity studies were carried out based on OECD guideline 423. The acute toxicity of 2 compounds, C25 and C34 was tested at the dose of 300 mg/kg. Both the compounds have not shown any clinical signs of toxicity. No mortality was observed by the test substance at the dose tested and found to be safer. Based on this result the novel compound C25was used at 10 and 20 mg/kg dose for in vivo efficacy evaluation.
Protective effect of novel glitazone compound C25
Results of the pharmacokinetic activity revealed that C25 has better kinetic activity when compared to C34. Hence, the C25 was taken for evaluation of anti-Parkinsonism activity on rotenone-induced PD mouse model at two different dose levels.
Parkinsonism was induced successfully in the mice by giving rotenone intraperitoneally for 21 days (2 mg/kg BW). As shown in Table 5, the control group animals have shown decreased locomotor activity significantly on days 14 and 21. The treatment with C25 has reversed the activity dose-dependently and similar to that of standard.
The same was observed in the rotarod behavioral studies (Table 6) done by employing a rotarod apparatus. There was a significant decrease in the time spent by the control group of animals on rotarod when compared with normal group animals, which signifies the induction of motor dysfunction by rotenone. When the C25 was administered alone with rotenone, the fall of time in rotarod was increased dose-dependently and was found as significant as that of standard drug.
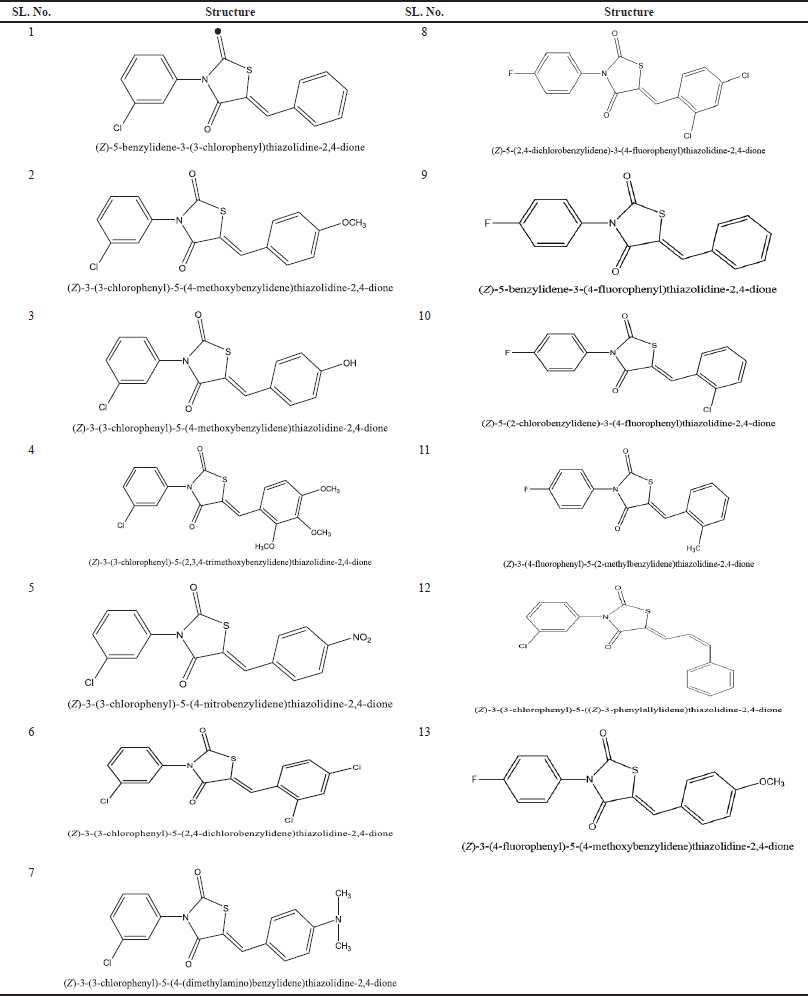 | Table 1. Structure of novel and virtual glitazones taken for docking studies. [Click here to view] |
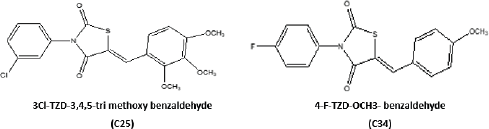 | Figure 1. Structure of glitazones C25 and C34 selected from docking studies. [Click here to view] |
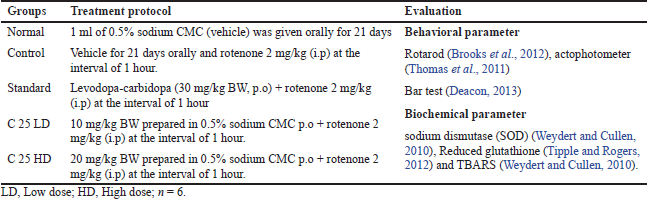 | Table 2. Evaluation of anti-Parkinson’s activity in rotenone induced animal model of Parkinson disease. [Click here to view] |
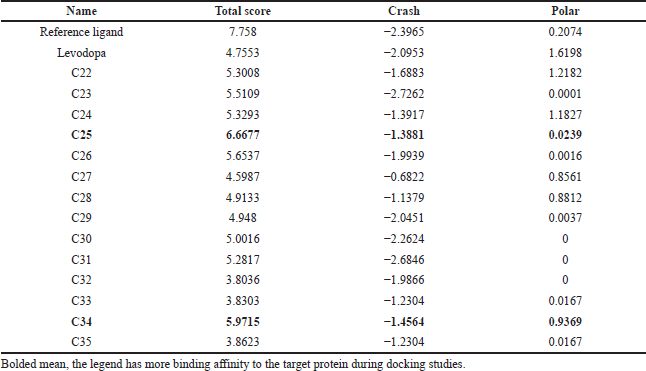 | Table 3. Results of molecular docking of novel virtual glitazone compounds. [Click here to view] |
Bar strength was the one more behavioral parameter employed to assess the activity of novel glitazone compound C25. As observed in the above parameters, control group showed a significant increase in the cataleptic activity on day 14 and day 21 when compared to the normal group which showed significant induction of PD by rotenone. There was a significant decrease in the cataleptic activity of treatment groups than the control group, which can be comparable with that of the standard group (Table 7).
Endogenous antioxidant enzymes
Rotenone induces PD mainly by increasing oxidative stress and causes mitochondrial complex I inhibition which leads to a decrease in the biogenesis of mitochondria and increases the level of reactive oxygen species that induced oxidases stress (Jollow et al., 1974).
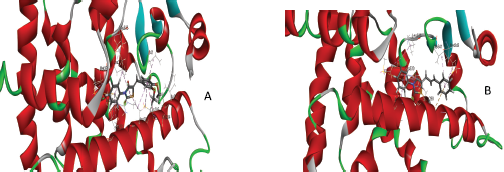 | Figure 2. Binding interaction of novel glitazone compound with the protein 3CS8 in 3D (after docking). A) C25 and B) C34. [Click here to view] |
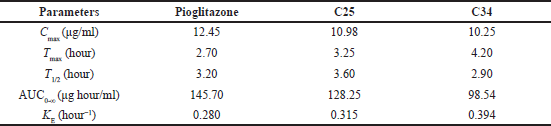 | Table 4. Pharmacokinetic data of the compounds C25 and C34. [Click here to view] |
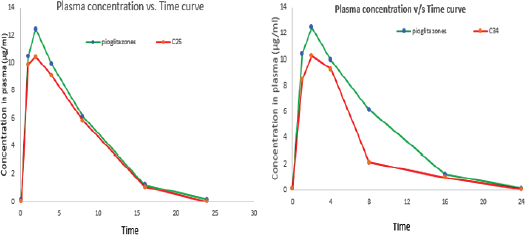 | Figure 3. Plasma concentration of C25 versus time and C34 versus time. [Click here to view] |
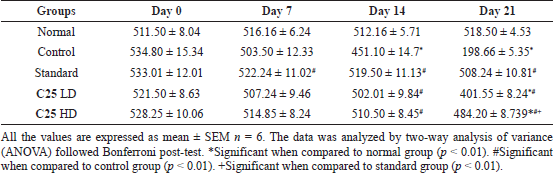 | Table 5. Anti-parkinsonism activity of novel glitazones derivatives on rotenone induced Parkinson activity in mice (locomotor activity). [Click here to view] |
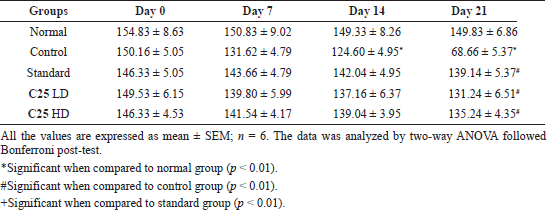 | Table 6. Anti-parkinsonism activity of novel glitazones derivatives on rotenone induced Parkinson activity in mice (grip strength by rotarod). [Click here to view] |
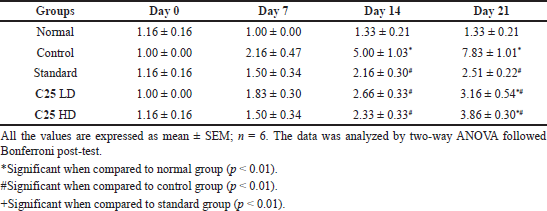 | Table 7. Anti-parkinsonism activity of novel glitazones derivatives on rotenone induced Parkinson activity in mice (catalepsy by bar test). [Click here to view] |
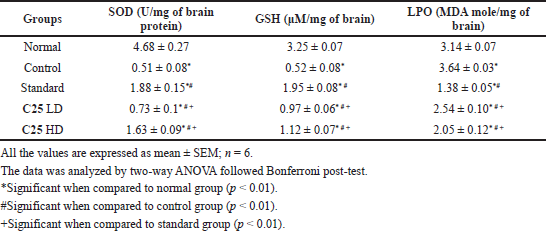 | Table 8. Anti-parkinsonism activity of novel glitazones derivatives on rotenone induced Parkinson activity in mice [SOD, Glutathione (GSH) and lipid peroxidation (LPO)]. [Click here to view] |
The effects of novel glitazone on endogenous antioxidant enzymes are given in Table 8. The animals treated alone with rotenone have shown decreased anti-oxidant enzymes, which suggests rotenone may induce oxidative stress. Novel glitazone compound has reversed the oxidative stress induced by rotenone.
DISCUSSION
The bioenergetics defects in neurodegeneration could be compensated by increased neuronal mitochondrial content. The important regulator of mitochondrial function in central nervous system is believed to be PPAR-γ co activator 1α (PGC-1α) which up regulates the biogenesis of mitochondria through Sirtuin 1 (Mahajan et al., 2016). PPAR-γ agonists like rosiglitazone, pioglitazone, and troglitazones which are commonly used for the management of diabetes were reported to increase mitochondrial biogenesis (Howitz et al., 2003). In another study, it was found that the PGC-1α agonist, like resveratrol and bezafibrate, have shown the protective effect against 6-hydroxydopamine in animal models (Khan et al., 2010). Studies conducted at our laboratory have shown that the novel glitazones have promising action against PD induced experimental animal model (Suma et al., 2019). In the present study, the novel glitazones were docked virtually with PGC-1 α protein to get best ligands and optimized legends were further tested in experimental animal model of PD.
The docking studies on novel glitazones (14 virtual ligands) to protein active sites were performed by an advanced molecular docking program SYBYL X 2.1.1. Scoring functions were used for determining the biding affinities of the ligands with the target receptors. The designed analogues were docked toward 3CS8 (target PGC-1α bound to PPAR-gamma receptor protein) in order to ascertain their binding ability to PPAR-gamma which in turn activity PGC-1 α protein (Justin et al., 2020).
The result had showed that the compounds C25 and C34 were having better binding activity with the target protein and were shown good polarity score and crash score than the other compounds when compared with the reference ligand (Mandal et al., 2018). The total score should be more for ligand to possess better binding affinity with the target receptor and crash score should be less. The compound C25 showed better binding affinity when compared to the compound C34 in the docking studies.
To assess the bioavailability of novel glitazones, a kinetic study was performed by single oral administration and the kinetic data were determined by the one compartment model. The experimental animals (Wistar Albino Rats) were administered a single oral dose of the compounds C25 and C34 and the kinetic data of the compounds are compared with the data of standard pioglitazone drug. The results showed that the C25 have better plasma concentration than C34 when compared with the standard pioglitazone compound. The plasma concentration of C25 was higher because the elimination rate of compound C34 was higher than the C25. The Tmax of the C25 was less than the C34 it signifies that C25 have the better absorption than the C34. The elimination rate of the C34 is more than the C25, so the plasma concentration is less than C25. The compound C25 shown better T1/2 than the C34 it signifies the duration of action of C25 is more than C34, it will retain in the plasma for more time and produce better action. The C25 also showed better T1/2 when compared to the standard pioglitazone. Based on the above studies C25 was chosen for further toxicity and efficacy studies in experimental animal model.
OECD guideline 423 was applied to know the acute toxicity studies of novel glitazone derivative C25, which comprises a 14-day observation studies (OECD guideline 423, 2001). Our findings indicate that the novel glitazone C25 has shown no mortality at 300 mg/kg dose tested for 14 days period. Then we considered this dose as tolerable dose and taken 10 and 20 mg/kg as test doses for efficacy study.
In the present study, we tried to examine the anti parkinsonian activity of novel glitazone on rotenone induced neurotoxicity in mice. The results of the study clearly demonstrate the beneficial role of novel glitazone C25 in attenuating the damage of dopaminergic neurons in PD induced mice. As discussed above, the PD is characterized by selective degeneration of dopaminergic neurons in substantia nigra. Pathogenesis of PD may not be known completely, the role of mitochondrial dysfunction, oxidative damage, and apoptosis are major contributing factors (Erba? et al., 2016). Rotenone induced neurotoxicity has been extensively employed as an animal model for PD. Since it is lipophilic in nature, it can enter organs like mitochondria and inhibits complex I activity of mitochondrial electron transport chain. This leads to development of ROS, adenosine tri phosphate reduction, and finally cell death in the neuron. Even the treatment with rotenone has demonstrated the histopathalogical changes as similar to PD (Cannon and Greenamyre, 2011; Xiong et al., 2009).
The results of the present study clearly indicate that rotenone administration could successfully develop PD like motor and behavioral features in rats such as hyperkinesias, flexed posture, and freezing. The compound C25 was assessed for its anti-parkinsonian activity at two different doses low dose (10 mg/kg) and high dose (20 mg/kg) and the activity was evaluated by behavioral parameters, like rotarod, spontaneous locomotor activity and cataleptic behavior by bar test.
Locomotor activity was assessed on day 7, 14, and 21, and the control group showed decrease in locomotor activity which confirms the induction of disease. The test drug increases the locomotor activity to the maximum of 95% and causes reversal rotenone neurotoxicity.
We observed decrease in fall of time by control group animals when tested by rotarod method, whereas the treatment with C25 has increased the fall of time dose dependently and maximum at higher doses. This demonstrates the loss of muscle coordination and muscular contraction in rotenone alone administered group animals and the same was attenuated by C25 administration.
We also found that C25 treated animals have shown decrease in the cataleptic activity when compared to control group animals dose dependently. Rotenone alone administered animals have taken more time to come back to the normal posture, which confirms the development of PD like symptom. This symptom was attenuated by administration of C25 and maximum effect was observed at higher dose tested.
As discussed above, rotenone induces PD mainly by increasing the oxidative stress, which causes mitochondrial complex I inhibition, leads to decrease in biogenesis of mitochondria. We observed elevated level of lipid peroxidation, decreased level of antioxidant enzymes SOD and GSH in brain tissues. Treatment with C25 lessened the level of lipid peroxidation and elevated the level of SOD and GSH.
Oxidative stress is one of the major reasons in many neurodegenerative diseases for the damage of nerve cells. In PD, oxidation of dopamine by mano amino oxidase-B and aldehyde dehydrogenase generates free radicals (OH) in presence of ferrous ions (basal ganglia rich in iron). Thus, the synthetic compounds possessing antioxidant activities appear to be the potential targets for developing new remedies in neuroprotection as the intake of antioxidants appears to be benefit in conditions of oxidative stress by maintaining the balance between the generation and scavenging of free radicals. The glitazone derivatives have a potent antioxidant and anti-PD activities.
CONCLUSION
The data obtained from this study reveals that novel glitazone compound C25 has protective effects on a rotenone-induced PD mouse model. The activity was dose-dependent and significant to the standard at higher dose tested. It is also exhibited significant antioxidant activity, which may additional advantages. But further studies are required to support the present assumption and elucidate the detailed neuroprotective mechanism.
AUTHOR CONTRIBUTIONS
All authors are contributed equally to the research work present in the manuscript.
ACKNOWLEDGMENT
The authors are grateful to Principal, JSS College of Pharmacy, Mysuru, and thanks to JSS Academy of Higher Education & Research, Mysuru for providing necessary facilities required for our research work.
CONFLICT OF INTEREST
The authors have no conflict of interest.
FINANCIAL SUPPORT
No funding received for the project and it is an M. Pharm dissertation project supported by JSS College of Pharmacy, JSS AHER, Mysuru.
REFERENCES
Alves G, Forsaa EB, Pedersen KF, Gjerstad MD, Larsen JP. Epidemiology of Parkinson’s disease. J Neurol, 2008; 255(5):18–32. CrossRef
Amin S, Al Okda A, Rashed L. Neuroprotective effects of piracetam versus peroxisome proliferator-activated receptor-gamma agonist pioglitazone in drug-induced Parkinsonism. Int Ann Med, 2017; 19:1–4. CrossRef
Brooks SP, Trueman RC, Dunnett SB. Assessment of motor coordination and balance in mice using the rotarod, elevated bridge and footprint tests. Curr Protoc Mouse Biol, 2012; 2:37–53. CrossRef
Cannon JR, Tapias V, Na HM, Honick AS, Drolet RE, Greenamyre JT. A highly reproducible rotenone model of Parkinson’s disease. Neurobiol Dis, 2009; 34(2):279–90. CrossRef
Cannon JR, Greenamyre JT. The role of environmental exposures in neurodegeneration and neurodegenerative diseases. Toxicol Sci, 2011; 124:225–50. CrossRef
Corona JC, Duchen MR. PPARγ and PGC-1α as therapeutic targets in Parkinson’s disease. Neurochem Res, 2015; 40:308–16. CrossRef
Deacon RMJ. Measuring motor coordination in mice. J Vis Exp, 2013; 29(75):2609–11. CrossRef
Erba? O, Y?lmaz M, Ta?k?ran D. Levetiracetam attenuates rotenone-induced toxicity: a rat model of Parkinson’s disease. Environ Toxicol Pharmacol, 2016; 42:226–30. CrossRef
Fujita Y, Yamada Y, Kusama M, Yamauchi T, Kamon J, Kadowaki T, Iga T. Sex differences in the pharmacokinetics of pioglitazone in rats. Comp Biochem Physiol Part C, 2003; 136(1):85–94. CrossRef
Hirsch EC, Hunot S. Neuroinflammation in Parkinson’s disease: a target for neuroprotection? Lancet Neurol, 2009; 8(4):382–97. CrossRef
Howitz KT, Bitternan KJ, Cohen HY. Small molecule activators of sirtuins extend Saccharomyces cerevisiae lifespan. Nature, 2003; 425:191–96. CrossRef
Hu Q, Wang G. Mitochondrial dysfunction in Parkinson’s disease. Transl Neurodegener, 2016; 5:14–5. CrossRef
Jollow DJ, Mitchell JR, Zampaglione N, Gillette JR. Bromobenzene-induced liver necrosis, the protective role of glutathione and evidence for 3,4-bromobenzene oxide as the hepatotoxic metabolite. Pharmacology, 1974; 11(3):151–69. CrossRef
Justin A, Mandal S, Prabitha P, Dhivya S, Yuvaraj S, Kabadi P, Sekhar SJ, Sandhya CH, Wadhwani AD, Divakar S, Bharathi JJ. Rational design, synthesis and in vitro neuroprotective evaluation of novel glitazones for PGC-1α activation via PPAR-γ: a new therapeutic strategy for neurodegenerative disorders. Neurotox Res, 2020; 37(3):508–24. CrossRef
Khan MM, Ahmad A, Ishrat T. Resveratrol attenuates 6-hydroxydopamine induced oxidative damage and dopamine depletion in rat model of Parkinson’s disease. Brain Res, 2010; 1328:139–51. CrossRef
Mahajan M, Deshmukh SN, Dudhgaonkar S. Novel therapies in the treatment of Parkinson’s disease. Int J Pharmacol Res, 2016; 6(01):1–6.
Mandal SP, Garg A, Prabitha P, Wadhwani AD, Adhikary L, Kumar BRP. Novel glitazones as PPARγ agonists: molecular design, synthesis, glucose uptake activity and 3D QSAR studies. Chem Cent J, 2018; 12:141–52. CrossRef
Moreno S, Farioli-vecchioli S, Ceru MP. Immunolocalization of peroxisome proliferator-activated receptors and retinoid X receptors in the adult rat CNS. Neuroscience, 2004; 123(1):131–45. CrossRef
Naim MJ, Alam O, Alam MJ, Hassan MQ, Siddiqui N, Naidu VGM, Alam MI. Design, synthesis and molecular docking of thiazolidinedione-based benzene sulphonamide derivatives containing pyrazole core as potential anti-diabetic agents. Bio Org Chem, 2018; 76:98–112.
OECD guidelines 423. OECD guideline for testing of chemicals acute oral toxicity—acute toxic class method. Available via https://ntp.niehs.nih.gov/iccvam/suppdocs/feddocs/oecd/oecd_gl423.pdf (Assessed 12 August 2018).
TBARS Estimation. Oxford Biomedical Research. Fluorometric microplate assay for 2-thiobarbituric acid reactive substances. Available via https://www.oxfordbiomed.com/sites/default/files/spec_sheet/FR45.pdf (Assessed 12 November 2018).
Pandit V, Pai RS, Devi K, Singh G, Narayana S, Suresh S. Development and validation of the liquid chromatographic method for simultaneous estimation of metformin, pioglitazone, and glimepiride in pharmaceutical dosage forms. Pharm Methods, 2012; 3(1):9–13. CrossRef
Rangaraj N, Vaghasiya K, Jaiswa S, Sharma A, Shukla M, Lal J. Do blood sampling sites affect pharmacokinetics? Chem Biol Interface, 2014; 4(3):176–91.
Schintu N, Frau L, Ibba M, Garau A, Carboni E, Carta AR. Progressive dopaminergic degeneration in the chronic MPTPp mouse model of Parkinson’s disease. Neurotox Res, 2009; 16(2):127–39. CrossRef
Straus DS, Glass CK. Anti-inflammatory actions of PPAR ligands: new insights on cellular and molecular mechanisms. Trends Immunol, 2007; 28(12):551–8. CrossRef
Suma BK, Krishna KL, Shetty AB, Nandini HS, Prashanth Kumar BR. Evaluation of anti-Parkinson’s activity of novel glitazones on rats. Int J Pharm Sci Rev Res, 2019; 55(2):64–9.
Thomas C, Marcaletti S, Feige JN. Assessment of spontaneous locomotor and running activity in mice. Curr Protoc Mouse Biol, 2011; 1:185–98. CrossRef
Tipple TE, Rogers LK. Methods for the determination of plasma or tissue glutathione levels. Methods Mol Biol, 2012; 889:315–24. CrossRef
Weydert CJ, Cullen JJ. Measurement of superoxide dismutase, catalase and glutathione peroxidase in cultured cells and tissue. Nat Protoc, 2010; 5(1):51–66. CrossRef
Xiong N, Huang J, Zhang Z, Zhang Z, Xiong J, Liu X. Stereotaxical infusion of rotenone: a reliable rodent model for Parkinson’s disease. PLoS One, 2009; 184:e7878.