INTRODUCTION
The use of traditional Chinese medicine (TCM) for the prevention and treatment of cancer has generated much interest. Recent monographs have documented more than 400 species of Chinese medicinal herbs associated with anti-cancer (Cai et al., 2004). Major classes of phenolic compounds found in these herbs include flavonoids, phenolic acids, tannins, coumarins, lignans, quinones, stilbenes, and curcuminoids.
Among the hundreds of medicinal herbs is Erigeron breviscapus, which is used in folk medicine for the treatment of paralysis, rheumatism, gastritis, toothache, and fever (Gao et al., 2017a; Wang and Ma, 2018). About 400 species of Erigeron have been documented in Asia, Europe, and North America, with 39 species in China, of which 14 are endemic and six are introduced (Chen and Brouillet, 2011).
Erigeron breviscapus (Vaniot) Handel-Mazzetti or Lamp Chrysanthemum belongs to the daisy family of Asteraceae (Chen and Brouillet, 2011; Gao et al., 2017a). It is a perennial herb that forms clumps of a meter in height. Flowers resemble those of daisies with central bright yellow disk florets surrounded by white ray florets at the periphery (Fig. 1). Each flower is actually an inflorescence known as capitulum. A traditional Chinese herb known as Deng Zhan Hua, E. breviscapus is found in Guangxi, Yunnan, Sichuan, Guizho, and Xizang provinces in the southwestern part of China, at altitudes of 1,200–3,600 masl. Three varieties of E. breviscapus have been recognized.
From the dried herbs of E. breviscapus, flavonoids (apigenin, apigenin-7-O-glucuronide, luteolin, scutellarein, and scutellarin) and phenolic acids (caffeic acid, chlorogenic acid, cynarin, isochlorogenic acid, and neochlorogenic acid) have been identified (Tian et al., 2017). Other classes of compounds include coumarins, glycosides, and essential oils. Scutellarin has also been reported in Scutellaria species (family Lamiaceae), such as S. baicalensis, S. lateriflora, and S. racemosa (Cole et al., 2008).
This review is focused on the chemistry and anti-cancer properties of scutellarin. Of particular interest are the multiple molecular targets and pathways of the compound toward a wide spectrum of human cancer cells, structure-activity relationships of cytotoxicity, and future research. References cited were procured from databases such as ScienceDirect, Google Scholar, and PubMed.
 | Figure 1. Plant (left) and flowers (right) of E. breviscapus. [Click here to view] |
SCUTELLARIN
Chemistry
Scutellarin (4’,5,6,7-tetrahydroxyflavone-7-O-glucuronide) is a flavone glycoside with a molecular formula of C21H18O12 and molecular weight of 462.35 g/mol (Gao et al., 2017a). The flavone is the glycone of scutellarein. The molecular structure of scutellarin comprises aromatic rings A and C that are fused together and attached to a phenyl ring B at position 2 of the ring C (Fig. 2). The molecule has an –OH group attached to C5, C6, and C4’, a glucuronide (–OGlu) moiety at C7, a carbonyl group at C4, and a C2–C3 double bond. Compounds with similar molecular structures as scutellarin are scutellarein with –OH group at C7, hispidulin with –OCH3 moiety at C6, and apigenin with –H group at C6 (Plochmann et al., 2007).
Besides scutellarin, other flavonoids isolated from E. breviscapus are apigenin, apigenin 7-O-glucuronide, baicalin, quercetin, quercetin-3-O-glucuronide, and scutellarein (Qu et al., 2001). Breviscapine is a crude extract of E. breviscapus which is used as TCM to improve blood circulation and cerebral blood supply (Gao et al., 2017a). The extract contains scutellarin (>85%) and apigenin-7-O-glucuronide as major components. Scutellarin has also been reported in other Scutellaria species such as S. altissima (Gao et al., 2017b), S. barbata (Zhang et al., 2003), S. multiradiatus (Luo et al., 2008), S. acer (Zhang et al., 2009b), and in the roots of S. baicalensis, another TCM plant with anti-cancer properties (Horvath et al., 2005).
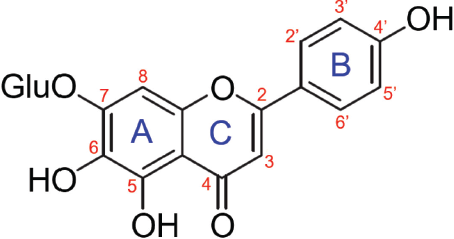 | Figure 2. Molecular structure of scutellarin. [Click here to view] |
Anti-cancer properties
There are two newly published reviews on the pharmacology of scutellarin by Wang and Ma (2018) and by Chledzik et al. (2018) that included its anti-cancer properties. Scientific evidence revealed that scutellarin possesses anti-cancer activities against a wide spectrum of cancer cells, including liver, prostate, lung, breast, colon, tongue, and renal cancers. Scutellarin induces apoptosis and cell cycle arrest and inhibits proliferation and progression of cancer cells via multiple molecular targets and pathways (Wang and Ma, 2018). Some molecular mechanisms of scutellarin-induced growth inhibition against different cancer cell lines are listed in Table 1.
Scutellarin induced apoptosis of liver cancer cells by down-regulating Bcl-2, Bax, and caspase-3, promising to be a useful anti-cancer drug candidate against liver cancer (Wu et al., 2010). Similarly, scutellarin reduced reactive oxygen species production and induced apoptosis of liver cancer cells by inhibiting the signal transducer and activator of transcription (STAT) 3 pathway, and transcriptional targets of Bcl-XL and Mcl-1 (Xu and Zhang, 2013). These findings were reaffirmed by a recent study that scutellarin suppressed migration and invasion of liver cancer cells by inhibiting the activities of STAT3, Girdin, and AKT (Ke et al., 2017).
Against prostate cancer cells, scutellarin suppressed cell proliferation by inducing apoptosis and promoting G2/M cell cycle arrest (Gao et al., 2017b), and by damaging DNA and by MCM protein-7 modulation (Guan et al., 2017). Against lung cancer cells, scutellarin inhibited cell growth and increased apoptosis by enhancing microRNA-7 (Zeng and Cai, 2017). Concurrently, another study reported that scutellarin increased apoptosis and autophagy of lung cancer cells via ERK/p53 and c-met/AKT signaling pathways (Sun et al., 2018).
Against breast cancer cells, scutellarin inhibited cell proliferation and invasion by up-regulating the Hippo/YAP signaling pathway (Hou et al., 2017). Scutellarin promoted apoptosis and metastasis of colon cancer cells through activation of caspase-6 (Chan et al., 2009) and through inhibition of ephrinb2 signaling (Zhu et al., 2017). Against tongue cancer cells, scutellarin inhibited the migration of cells by regulating the production of αvβ6 integrin and E-cadherin (Li et al., 2010) and suppressed proliferation and induced apoptosis of cells by inhibiting MMP-2 and -9 expression (Li et al., 2013). In addition, scutellarin suppressed proliferation and induced apoptosis of cervical cancer cells by inhibiting the activity of pyruvate kinase M2 (You et al., 2017). Against renal cancer cells, scutellarin inhibited cell proliferation and migration via up-regulation of phosphatase and tensin homolog, and inhibition of P13K/AKT/mTOR signaling (Deng et al., 2018). Finally, scutellarin has been reported to induce apoptosis of B-lymphoma cells via activation of caspases (Feng et al., 2012).
 | Table 1. Molecular mechanisms of scutellarin-induced growth inhibition against different cancer cell lines. [Click here to view] |
SAR and cytotoxicity
A study on structure-activity relationships (SAR) conducted on cytotoxicity of 25 flavonoids against Jurkat E6-1 human leukemia cells showed that the cytotoxicity (EC50) of scutellarin (44 μM) was 11.4 times stronger than that of scutellarein (502 μM) (Plochmann et al., 2007). Scutellarin ranked second, while scutellarein ranked eighteenth. It is evident that glucuronidation at C7 of scutellarin (Fig. 2) significantly increased its cytotoxicity. Scutellarin with triple hydroxylation at C5, C6, and C4’, a C2–C3 double bond, and a C4 carbonyl group was more than 50 times more cytotoxic than taxifolin, without these structural components but an –OH group at C3 (Plochmann et al., 2007).
MCF-7 breast cancer cells cultured with different concentrations of scutellarin in vitro were analyzed for anti-proliferation and apoptosis of cells (Hou et al., 2017). Results showed that the inhibition rates of proliferation were 40%, 60%, and 70% for 24, 48, and 72 hours, respectively. Apoptotic cells ranged from 12.4% to 24.0% in 40–120 μM scutellarin groups, compared with 7.8% in the control group.
Results of another recent study showed that the cytotoxicity of scutellarin had IC50 values of 77, 73, and 56 μM against HCT-116, PC-3, and HepG-2 cancer cells, respectively (Han et al., 2017). Cytotoxicity was not observed against MCF-7 and L-O2 cancer cells. In the experiment, most of the derivatives of scutellarin synthesized with benzyl groups displayed significantly stronger cytotoxicity, including MCF-7 and L-O2 cancer cells.
Other properties
Besides anti-anticancer activities, the other pharmacological properties of scutellarin include antioxidant (Guo et al., 2011), neuroprotection (Guo et al., 2011; Wang et al., 2011), cardioprotection (Gao et al., 2017a), anti-inflammation (Wang et al., 2011), anti-diabetic (Long et al., 2015), anti-obesity (Lu et al., 2013), and anti-HIV (Zhang et al., 2005). Scutellarin also provides protection against disorders such as myocardial infarction (Huang et al., 2018), neuroinflammation (You et al., 2018; Yuan et al., 2016), hypertension (Chen et al., 2013), hypercholesterolemia (Li et al., 2009), cardiotoxicity (Sun et al., 2017), and cerebral ischemia (Lin et al., 2007; Zhang et al., 2009a).
CONCLUSION
Scutellarin possesses potent anti-cancer properties with great therapeutic potentials. However, some disadvantages limit its application due to low stability and poor oral bioavailability. Derivatives synthesized from a chemical modification of scutellarin can be developed into anti-tumor agents with high efficiency and low toxicity. SAR studies would be useful in the analysis of derivatives with enhanced cytotoxicity. Likewise, the use of scutellarin in combination with other drugs may yield better results. Future directions can also focus on human population studies and on clinical trials to evaluate the safety and efficacy of scutellarin in the prevention and treatment of cancer.
ACKNOWLEDGMENTS
We would like to acknowledge UCSI University (Proj-In-FAS-049; Proj-In-FAS-052) and the Ministry of Higher Education, Malaysia (FRGS/2/2014/SG01/UCSI/02/2) for supporting our research group.
REFERENCES
Cai YZ, Sun M, Xing J, Luo Q, Corke, H. Structure-radical scavenging activity relationships of phenolic compounds from traditional Chinese medicinal plants. Life Sci, 2005; 74:2157−84.
Chan JY, Tan BKH, Lee SC. Scutellarin sensitizes drug-evoked colon cancer cell apoptosis through enhanced caspase-6 activation. Anticancer Res, 2009; 29:3043−8.
Chen X, Shi X, Zhang X, Lei H, Long S, Su H, Pei Z, Huang R. Scutellarin attenuates hypertension-induced expression of brain toll-like receptor 4/nuclear factor kappa B. Mediators Inflamm, 2013; Article ID 432623, 9 p.
Chen Y, Brouillet L. Erigeron L. Flora China, 2011; 20–21:635−8.
Chledzik S, Strawa J, Matuszek K, Nazaruk J. Pharmacological effects of scutellarin, an active component of genus Scutellaria and Erigeron: a systematic review. Am J Chin Med, 2018; 46:319−37.
Cole IB, Cao J, Alan AR, Saxena PK, Murch SJ. Comparisons of Scutellaria baicalensis, Scutellaria lateriflora and Scutellaria racemosa: genome size, antioxidant potential and phytochemistry. Planta Med, 2008; 74:1−8.
Deng W, Han W, Fan T, Wang X, Cheng Z, Wan B, Chen J. Scutellarin inhibits human renal cancer cell proliferation and migration via up-regulation of PTEN. Biomed Pharmacother, 2018; 107:1505−13.
Feng Y, Zhang S, Tu J, Cao Z, Pan Y, Shang B, Liu R, Bao M, Guo P, Zhou Q. Novel function of scutellarin in inhibiting cell proliferation and inducing cell apoptosis of human Burkitt lymphoma Namalwa cells. Leuk Lymphoma, 2012; 53:2456−64.
Gao J, Chen G, He H, Liu C, Xiong X, Li J, Wang J. Therapeutic effects of breviscapine in cardiovascular diseases: a review. Front Pharmacol, 2017a; 8:289.
Gao C, Zhou Y, Jiang Z, Zhao Y, Zhang D, Cong X, Cao R, Li H, Tian W. Cytotoxic and chemo-sensitization effects of scutellarin from traditional Chinese herb Scutellaria altissima L. in human prostate cancer cells. Oncol Rep, 2017b; 38:1491−9.
Guan YB, Yang DR, Nong SJ, Ni J, Hu CH, Li J, Zhu J, Shan YX. Breviscapine (BVP) inhibits prostate cancer progression through damaging DNA by mini-chromosome maintenance protein-7 (MCM-7) modulation. Biomed Pharmacother, 2017; 93:103−16.
Guo H, Hu LM, Wang SX, Wang YL, Shi F, Li H, Liu Y, Kang LY, Gao XM. Neuroprotective effects of scutellarin against hypoxic-ischemic-induced cerebral injury via augmentation of antioxidant defense capacity. Chin J Physiol, 2011; 54:399−405.
Han T, Li J, Xue J, Li H, Xu F, Cheng K, Li D, Li Z, Gao M, Hua H. Scutellarin derivatives as apoptosis inducers: design, synthesis and biological evaluation. Eur J Med Chem, 2017; 135:270−81.
Horvath CR, Martos PA, Saxena PK. Identification and quantification of eight flavones in root and shoot tissues of the medicinal plant Huang-Qin (Scutellaria baicalensis Georgi) using high-performance liquid chromatography with diode array and mass spectrometric detection. J Chromatogr A, 2005; 1062:199−207.
Hou L, Chen L, Fang L. Scutellarin inhibits proliferation, invasion, and tumorigenicity in human breast cancer cells by regulating Hippo-YAP signaling pathway. Med Sci Monit, 2017; 23:5130−6.
Huang H, Geng Q, Yao H, Shen Z, Wu Z, Miao X, Shi P. Protective effect of scutellarin on myocardial infarction induced by isoprenaline in rats. Iranian J Basic Med Sci, 2018; 21:267−76.
Ke Y, Bao T, Wu X, Tang H, Wang Y, Ge J, Fu B, Meng X, Chen L, Zhang C, Tan Y. Scutellarin suppresses migration and invasion of human hepatocellular carcinoma by inhibiting the STAT3/Girdin/Akt activity. Biochem Biophy Res Commun, 2017; 483:509−15.
Li H, Huang D, Gao Z, Lv Y, Zhang L, Cui H, Zheng J. Scutellarin inhibits cell migration by regulating production of αvβ6 integrin and E-cadherin in human tongue cancer cells. Oncol Rep, 2010; 24:1153−60.
Li H, Huang D, Gao Z, Chen Y, Zhang L, Zheng J. Scutellarin inhibits the growth and invasion of human tongue squamous carcinoma through the inhibition of matrix metalloproteinase-2 and -9 and alphavbeta6 integrin. Int J Oncol, 2013; 42:1674−81.
Li Q, Wu J, Guo D, Cheng H, Chen S, Chan SW. Suppression of diet-induced hyper-cholesterolemia by scutellarin in rats. Planta Med, 2009; 75:1203−8.
Lin LL, Liu AJ, Liu JG, Yu XH, Qin LP, Su DF. Protective effects of scutellarin and breviscapine on brain and heart ischemia in rats. J Cardiovasc Pharmacol, 2007; 50:327−32.
Long L, Wang J, Lu X, Xu Y, Zheng S, Luo C, Li Y. Protective effects of scutellarin on type II diabetes mellitus-induced testicular damages related to reactive oxygen species/Bcl-2/Bax and reactive oxygen species/microcirculation/staving pathway in diabetic rat. J Diabetes Res, 2015; Article ID 252530:11.
Lu K, Han M, Ting HL, Liu Z, Zhang D. Scutellarin from Scutellaria baicalensis suppresses adipogenesis by up-regulating PPARα in 3T3-L1 cells. J Nat Prod, 2013; 76:672−8.
Luo P, Tan ZH, Zhang ZF, Zhang H, Liu XF, Mo ZJ. Scutellarin isolated from Erigeron multiradiatus inhibits high glucose-mediated vascular inflammation. J Pharm Soc Jpn, 2008; 128:1293−9.
Plochmann K, Korte G, Koutsilieri E, Richling E, Riederer P, Rethwilm A, Schreier P, Scheller C. Structure-activity relationships of flavonoid-induced cytotoxicity on human leukemia cells. Arch Biochem Biophy, 2007; 460:1−9.
Qu J, Wang Y, Luo G, Wu Z. Identification and determination of glucuronides and their aglycones in Erigeron breviscapus by liquid chromatography-tandem mass spectrometry. J Chromatogr A, 2001; 928:155−162.
Sun CY, Zhu Y, Li XF, Wang XQ, Tang LP, Su ZQ, Li CY, Zheng GJ, Feng B. Scutellarin increases cisplatin-induced apoptosis and autophagy to overcome cisplatin resistance in non-small cell lung cancer via ERK/p53 and c-met/AKT signaling pathways. Front Pharmacol, 2018; 9:92.
Sun XP, Wan LL, Yang QJ, Huo Y, Han YL, Guo C. Scutellarin protects against doxorubicin-induced acute cardiotoxicity and regulates its accumulation in the heart. Arch Pharm Res, 2017; 40:875−83.
Tian Y, Li Q, Zhou X, Pang Q, Xu Y. A UHPLC-MS/MS method for simultaneous determination of twelve constituents from Erigeron breviscapus extract in rat plasma: application to a pharmacokinetic study. J Chromatogr B, 2017; 1046:1−12.
Wang L, Ma Q. Clinical benefits and pharmacology of scutellarin: a comprehensive review. Pharmacol Ther, 2018; 190:105−27.
Wang S, Wang H, Guo H, Kang L, Gao X, Hu L. Neuroprotection of scutellarin is mediated by inhibition of microglial inflammatory activation. Neuroscience, 2011; 185:150−60.
Wu Y, Fan Q, Lu N, Tao L, Gao Y, Qi Q, Guo Q. Breviscapine-induced apoptosis of human hepatocellular carcinoma cell line HepG2 was involved in its anti-tumour activity. Phytother Res, 2010; 24:1188−94.
Xu H, Zhang S. Scutellarin-induced apoptosis in HepG2 hepatocellular carcinoma cells via a STAT3 pathway. Phytother Res, 2013; 27:1524−8.
You L, Zhu H, Wang C, Wang F, Li Y, Li Y, Wang Y, He B. Scutellarin inhibits HeLa cell growth and glycolysis by inhibiting the activity of pyruvate kinase M2. Bioorg Med Chem Lett, 2017; 27:5404−8.
You P, Fu S, Yu K, Xia Y, Wu H, Yang Y, Ma C, Liu D, Chen X, Wang J, Ye X. Scutellarin suppresses neuro-inflammation via the inhibition of the AKT/NF-κB and p38/JNK pathway in LPS-induced BV-2 microglial cells. Naunyn-Schmiedeberg’s Arch Pharmacol, 2018; 391:743−51.
Yuan Y, Fang M, Wu CY, Ling EA. Scutellarin as a potential therapeutic agent for microglia-mediated neuro-inflammation in cerebral ischemia. Neuromol Med, 2016; 18:264−73.
Zeng J, Cai S. Breviscapine suppresses the growth of non-small cell lung cancer by enhancing microRNA-7 expression. J Biosci, 2017; 42:121−9.
Zhang GH, Wang Q, Chen JJ, Zhang XM, Tam SC, Zheng YT. The anti-HIV-1 effect of scutellarin. Biochem Biophy Res Commun, 2005; 334:812−6.
Zhang HF, Hu XM, Wang LX, Xu SQ, Zeng FD. Protective effects of scutellarin against cerebral ischemia in rats: evidence for inhibition of the apoptosis-inducing factor pathway. Planta Med, 2009a; 75:121−6.
Zhang W, Di D, Wen B, Liu X, Jiang S. Determination of scutellarin in Scutellaria barbata extract by liquid chromatography-electrochemical detection. J Liq Chromatogr Rel Technol, 2003; 26:2133−40.
Zhang ZF, Wang JG, An J, Zhang H. Determination of scutellarin in Erigeron acer from different habitats by RP-HPLC. West China J Pharm Sci, 2009b; 1:36.
Zhu PT, Mao M, Liu ZG, Tao L, Yan BC. Scutellarin suppresses human colorectal cancer metastasis and angiogenesis by targeting ephrinb2. Am J Transl Res, 2017; 9:5094−104.