INTRODUCTION
A high-fat diet (HFD) is a profound detrimental factor promoting metabolic diseases including type 2 diabetes mellitus (T2DM) (Lozano et al., 2017; Skovsø et al., 2014); one of the life-threatening global health issues that is yet to be solved (Pearson, 2019). However, foods with high-fat content as found in various traditional and modern cuisines are inevitably palatable, thus being frequently consumed excessively by many people all over the world. Consequently, the prevalence of T2DM and associated metabolic disorders including obesity, cardiovascular, and fatty liver diseases is markedly soaring (American Diabetes Association, 2019; Zaharia et al., 2019). It is predicted that by 2045, the number of adults with diabetes will globally increase to 693 million, with T2DM accounting for the major part of the cases (Saklayen, 2019).
It has been demonstrated that HFD induces the development of T2DM by involving various mechanisms, including gut microbiota dysbiosis (Puddu et al., 2014), low-grade inflammation in both central and peripheral systems (Burhans et al., 2018; Gao et al., 2015), and dyslipidemia and lipid peroxidation (Estrany et al., 2011). Moreover, consumption of rich fiber diets has been strongly attributed to an improvement in plasma lipid profiles, reduction of body weight, and a sustained normal blood glucose level (Cantero et al., 2017; Guo et al., 2016; Han et al., 2019; Santoso et al., 2019). A study using mice fed with HFD revealed that the incorporation of dietary fibers such as sugarcane fiber and psyllium could effectively enhance insulin sensitivity, halt the excessive body weight increase, and modulate the secretion of metabolic hormones, particularly glucagon-like peptide 1 (GLP-1) and leptin (Wang et al., 2012). Another study investigating dietary fiber of bamboo shoots also found counteractive effects of dietary fiber against diet-induced metabolic diseases’ development (Li et al., 2016). Although the beneficial effects of plant-based resources of dietary fibers have been explored, to our knowledge, studies focusing on the edible fibers of the tuberous plants remain limited. One of the potential tuberous plants known for its nutrient contents and medicinal benefits against metabolic diseases is jicama (P. erosus, Fabaceae) (Buckman et al., 2017; Noman et al., 2012). In many Asian countries, including Indonesia, jicama tuber is usually consumed as juices and pickles or after being processed as cookies and chips. Many studies have revealed that the jicama juice and extract could effectively lower blood glucose levels and reduce the risk of cardiovascular diseases (Park and Han, 2015; Park et al., 2016; Thaptimthong et al., 2016). However, whether dietary fiber of jicama is also capable of eliciting the preventive and therapeutic effects against diet-induced metabolic diseases, particularly T2DM, remains unclear.
Our previous study on mice has revealed a beneficial effect of consuming jicama fiber against pancreatic adiposity and dystrophy of the islet of Langerhans caused by a high-sugar diet (HSD) (Santoso et al., 2020b). However, HSD is known to directly promote the glucotoxicity-induced pancreatic deterioration, wherin the development of T1DM (type 1 diabetes mellitus) that is mechanistically different from the HFD-induced T2DM development (Eizirik et al., 2020; Long et al., 2017). Accordingly, a counteractive effect elicited by jicama fiber supplementation could plausibly be different under the HFD challenge as compared with those observed in the HSD treatment. Hence, in this research, we aimed to clarify whether the jicama fiber is capable of precluding the dysregulated blood glucose profiles and pathological alterations in the pancreatic tissues, as indicators of T2DM development, promoted by HFD. We deployed an animal experimental study using adult healthy male mice as animal models fed with HFD. Our findings elucidate an effective and affordable way to diminish diet-induced T2DM development by consuming dietary fibers of jicama tuber.
MATERIALS AND METHODS
Provision of experimental animals
This study used healthy BALB (Bagg and Albino)/c mice (male, 8 weeks old, bodyweight 20–23 g). All individuals were subjected to acclimatization in the animal room for a week with a 12 h light–dark cycle and room temperature at 25°C–26°C. A normal chow diet (BP2; standard commercial diet for rodents, Java Comfeed) and tap water were provided ad libitum. All procedures for care and use of animals in this study were in accordance with the standard guideline established by the Committee of Research Ethics and Regulation of Andalas University, Indonesia (Approval number: T/15/UN.16.17/PT.01.03/PD-KO/2019, March 11, 2019).
Fiber extraction
The tubers of jicama were obtained from the local market in Padang city (West Sumatra Province) and washed with tap water four times before being peeled and grated using an electric grater machine (SENCOR SSG-3501 GR, Czech). Furthermore, the sample was subjected to fiber extraction following the protocol as described previously (Kumalasari et al., 2014a). Briefly, the grated sample was soaked in the dH2O for 18 hours at 4°C in a refrigerator (Thermofisher Scientific 288R-AEV-TS, USA); then, the supernatant was collected using a filter before being steamed for 1 hour at 100°C in automatic steamer (PHILIPS HD-9104, Netherland). The fibers were dried in an oven (Emmert UN55, Germany) for 17 hours at 66°C–68°C and ground to be powder using electric grinder (TENCAN Model XQM-0.4 A, China).
Experimental treatments
After acclimatization, the mice were randomly grouped into four different groups consisting of 10 individuals in each group. Furthermore, the mice were subjected to diet treatments as follows:
Group 1: fed with a normal diet (ND).
Group 2: fed with a HFD.
Group 3: fed with HFD + jicama fiber 10% (HFD + JF10%).
Group 4: fed with HFD + jicama fiber 25% (HFD + JF25%).
The treatments were deployed for 8 weeks. The ND was a standard commercial diet for rodents (BP2, Java Comfeed Indonesia) and HFD was a commercial fatty diet for rodents (CLEA, Shizuoka, Japan). The doses of fiber (10% and 25%) used in this experiment were decided based on our previous study (Santoso et al., 2019), which were considered as effective doses in precluding the HSD-induced diabetes development in mice.
Blood glucose measurement
A random blood glucose level was monitored once every 2 weeks at 9.00 am since the beginning of the treatment. Firstly, a topical anesthetic 0.75% bupivacaine (Kalbe Farma, Indonesia) was applied to the tip of the tail for 1–2 minutes before being cut with sterile surgical scissors. Furthermore, a drop of blood sample was drawn from the tip of the tail with minimum injury and the blood glucose level was subsequently determined using an automated glucometer (Glucocard, Takara, Japan). The fasting blood glucose was measured at the end of treatment after 18 hours fasting (food was deprived while drinking water was provided in the cage during fasting) using glucometer.
Glucose tolerance test (GTT)
The GTT was carried out at the end of treatment. Briefly, 3 days after fasting blood glucose measurement, the animals fasted for 6 hours (07:30 am−01:30 pm). Furthermore, the animals were injected intraperitoneally (i.p.) with glucose solution (Sigma-Aldrich, Merck Darmstadt, Germany) at a dose of 2 g/kg BW (bodyweight), followed by the blood glucose measurement at 0, 15, 30, 60, 90, and 120 minutes after injection using a glucometer (Glucocard, Takara, Japan).
Histopathological examination of pancreas
The animals were sacrificed a day after GTT and the pancreas tissue was collected immediately and fixed in 10% formaldehyde (Sigma-Aldrich, Merck Darmstadt, Germany) for 24 hours. Furthermore, the fixed sample was sent for histopathological examination by following the protocol of paraffin-embedded tissue processing (Elkotby et al., 2017). Briefly, the fixed tissues were dehydrated using graded ethanol (Pro Analis Merck 1.00983.250 Millippore, Danvers, MA) and then cleared in xylene (Sigma-Aldrich, Merck Darmstadt, Germany) before being embedded in paraffin (Paraplast, Sigma-Aldrich, Merck Darmstadt, Germany). Furthermore, the samples were cut using a rotary microtome (Leica RM2125RTS, Leica Biosystem, Buffalo Grove, IL) and stained with Hematoxylin and Eosin (TissuePro Technology EY07-500R, H08-500R, Gainesville, FL). The histological slides of the pancreas were examined under a microscope and the representative features of every slide were photographed using an integrated camera for microscope (Olympus CX31, Tokyo, Japan). The size of the islet of Langerhans and the total cell number in the islet was calculated using ImageJ software (obtained from the National Institute of Health, Bethesda, MD; https://imagej.nih.gov/ij/download.html). The pancreatic adiposity and fibrosis were observed by examining the occurrence of adipose tissue and fibrous connective tissue, respectively, in the pancreas.
Statistical analysis
All quantitative data are depicted as mean ± SE. An analysis of variance, followed by a Bonferroni post-hoc test was carried out to determine the significant difference among groups with p < 0.05, which was considered as significant.
RESULTS AND DISCUSSION
Effect of jicama fiber on blood glucose
A random blood glucose monitoring carried out every 2 weeks revealed that HFD increased the blood glucose level of mice since the second week of treatment until the end of treatment (Fig. 1A). On the contrary, the supplementation of jicama fiber at the dose 10% and 25% in HFD could sustain the normal blood glucose level along the time course of treatment. Statistical analysis indicated that the random blood glucose levels of the HFD group are significantly higher as compared with the jicama fiber’s treated groups and ND group (p < 0.05).
The measurement of fasting blood glucose level at the end of treatment showed a marked increase in the HFD group, while it was significantly lower in the jicama fiber-treated groups and ND group (p < 0.05, Fig. 1B). However, only jicama fiber at the dose of 25%, but not 10%, could sustain fasting blood glucose level to be comparable with the ND group (p > 0.05).
Effect of jicama fiber on glucose tolerance
The GTT test conducted at the end of treatment exhibited a glucose intolerance in the HFD group indicated by a significant increase of the blood glucose level particularly at the 15th and 30th minutes after injection of glucose (Fig. 2A, p < 0.05) and a markedly higher area under curve (AUC) (Fig. 2B, p < 0.05) as compared with the other groups. Moreover, glucose tolerance was sustained to be comparable with the ND treated group in the jicama fiber-treated groups (10% and 25%; p > 0.05).
Effect of jicama fiber on the histopathological alteration of the pancreas
A microscopic observation on the pancreatic tissue revealed that mice of HFD group exhibited an enlargement of the islet of Langerhans, indicating hypertrophy (Fig. 3B), while such enlargement was prevented in the jicama fiber-treated groups (10% and 25%) as well as ND group (Fig. 3A, C, and D). Furthermore, a measurement of the islet size revealed that the islets were significantly larger in the HFD group as compared with the jicama-treated groups and ND group (p < 0.05, Fig. 3E). Likewise, counting on total cell number in the islet of Langerhans found that it was significantly higher in the HFD group as compared with the jicama-treated groups and ND group (p < 0.05, Fig. 3F), suggesting the HFD-induced hyperplasia.
An investigation on the pancreatic adiposity revealed that the ectopic fat deposition in the pancreatic tissue was abundant in the HFD group (Fig. 4B) but not in the jicama-treated groups (Fig. 4C and D) and the ND group (Fig. 4A). Moreover, the fibrous connective tissue was observed in the HFD group (Fig. 4B) but absent in the jicama fiber groups and ND group.
Our present results demonstrate a preventive effect of dietary fiber of jicama tuber against the development of T2DM caused by HFD in mice. A previous study in rats showed that the excessive intake of fatty diet has been attributed to hyperglycemia, glucose intolerance, and insulin resistance, suggesting the development of T2DM (Lozano et al., 2017). Similarly, our study found that HFD treatment induced a hyperglycemic state and glucose intolerance, but it could be effectively precluded by the supplementation of jicama fiber. Moreover, our current data also demonstrated that the pancreatic tissue was protected from HFD-induced islet hyperplasia and hypertrophy in mice treated with jicama fiber. It has been reported that chronic insulin resistance caused by HFD is closely associated with a marked increase in insulin demand to overcome an apparent blood glucose elevation (Nagy et al., 2018). Such a condition could subsequently promote hypertrophy and hyperplasia of the pancreatic tissue, particularly the islet β cells, to overcome the excessive insulin demand (Golson et al., 2010). In our present study, neither plasma insulin level nor insulin tolerance was examined to clarify our suggestion that the supplementation of jicama fiber is capable of preventing HFD-induced insulin resistance. However, the consistency in normal blood glucose level and glucose tolerance as well as unaltered histological features of the islet of Langerhans observed in the jicama fiber-treated groups could be considered as evidence to support such consideration.
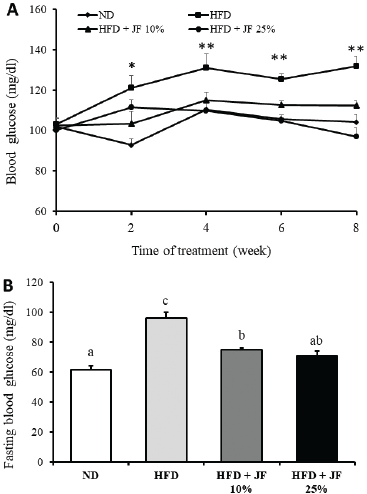 | Figure 1. Effect of jicama fiber on blood glucose profiles. (A) Random blood glucose measured every 2 weeks. (B) Fasting blood glucose measured at the end of treatment. ND: normal diet, HFD: high-fat diet, and JF: jicama fiber. n =10 for each group. Different characters above the bars represent the statistical difference among groups (p < 0.05). [Click here to view] |
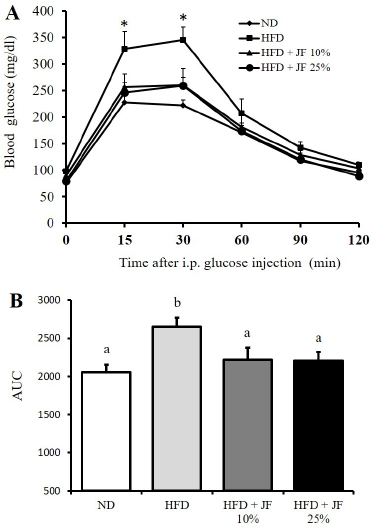 | Figure 2. Effect of jicama fiber on glucose tolerance. (A) Blood glucose level determined at different time points (in minutes) after an i.p. injection of glucose (2 g/kg BW) in GTT carried out at the end of treatment. (B) AUC calculated based on the blood glucose levels as measured in GTT. Different characters above the bars represent the statistical difference among groups (p < 0.05). [Click here to view] |
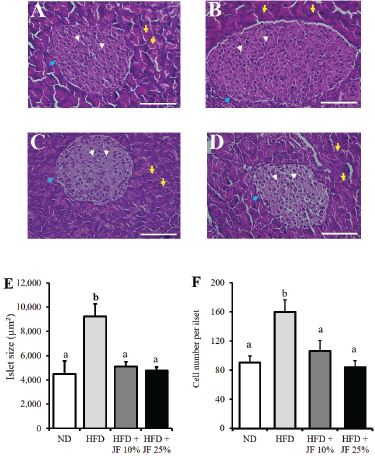 | Figure 3. Effect of jicama fiber on the histopathological alteration of pancreas. Representative photographs of the islet of Langerhans stained with HE in mice fed with (A) ND, (B) HFD, (C) HFD + JF 10%, (D) HFD + JF 25%, (E) the average size of the islet, and (F) average cell number per islet. In A–D, white arrows indicate the cells of islet of Langerhans; blue arrows indicate the edge of islet of Langerhans; and yellow arrows indicate acinar cells of pancreas; scale bars = 40 µm. Different characters above the bars in E and F represent the significant differences among groups (p < 0.05). [Click here to view] |
In addition to insulin resistance, other detrimental effects of the HFD promoting the development of diabetes have also been reported including those associated with ectopic adiposity and fibrosis of the pancreatic tissues (Matsuda et al., 2014; Ye et al., 2018; Yu et al., 2014). In our study, the adipocytes were abundantly observed in the pancreas of the HFD-fed mice, while the fibrous connective tissue indicating fibrosis was subtle. Furthermore, both pancreatic adiposity and fibrosis caused by HFD were absent in the jicama fiber-treated groups at both a lower dose (10%) and a higher dose (25%), suggesting the affectivity of the fiber in counteracting fat accumulation and tissue degeneration in the pancreas. Our previous study also revealed that jicama fiber exerted a beneficial effect against pancreatic adiposity and necrosis of the pancreatic acinar cells in mice fed with HSD particularly at a higher dose (25%) (Santoso et al., 2020b). We also have reported that jicama fiber at the dose of 25% could diminish fat deposition in the liver of HSD-treated mice (Santoso et al., 2020a). The ectopic adiposity in the pancreas is profoundly associated with the upregulation of cytokines that could lead to a severe inflammation, thereby cellular damages of the pancreas (Matsuda et al., 2014; Patel and Patel, 2015). Therefore, the counteractive effect of jicama fiber against pancreatic adiposity and fibrosis could alleviate the development of T2DM caused by HFD.
A large prospective cohort study revealed that the consumption of dietary fiber from cereal and whole grains could lower the risk of T2DM in humans (de Munter et al., 2007). Some plausible mechanisms underlying the preventive effect of dietary fibers against the development of metabolic diseases including T2DM have been proposed. The dietary fibers are capable of reducing the enzymatic digestion of nutrients including fat and carbohydrates in the gastrointestinal tract (Dhingra et al., 2012; Pouyamanesh et al., 2016). Consequently, it could minimize the loading of triglycerides, fatty acids, and glucose into the circulatory systems, thereby mitigating further detrimental effects of having the excessive intake of high-energy diet. The dietary fibers are also capable of inducing the production of hormone, namely GLP-1 in the intestinal L-cells (Bodnaruc et al., 2016). An increase in GLP-1 release is profoundly implicated in slowing gastric emptying rate that could further delay food digestion and nutritional absorption (Nadkarni et al., 2014). Moreover, GLP-1 plays a pivotal role in the blood glucose homeostasis by its action in enhancing insulin secretion and reducing glucagon production in the pancreas (Larraufie et al., 2019). However, it remains to be investigated in the future whether jicama fiber is also capable of inducing GLP-1 secretion to subsequently alleviate the HFD effect on the metabolic homeostasis.
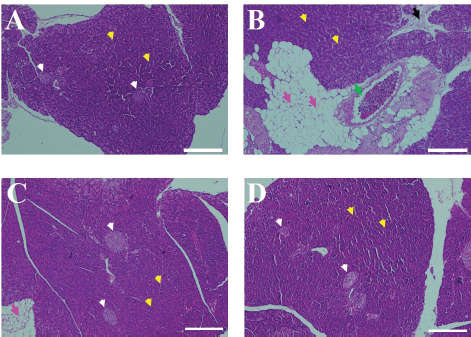 | Figure 4. Effect of jicama fiber on ectopic fat deposition and fibrosis in the pancreatic tissue. The representative photographs of pancreas in mice fed with (A) ND, (B) HFD, (C) HFD + JF 10%, and (D) HFD + JF 25%. In A–D, white arrows indicate the islet of Langerhans; yellow arrows indicate acinar cells of pancreas; pink arrows indicate adipose tissue (ectopic fat deposition); black arrow indicates fibrous connective tissue (fibrosis); and green arrow indicates blood vessel; scale bars = 100 µm. [Click here to view] |
A proper intake of dietary fiber has been associated with an increase in the level of short-chain fatty acids (SCFAs) which resulted from the microbial fermentation of fibers in the gut (Zhai et al., 2018a, 2018b). Among various kinds of SCFAs, the acetate, propionate, and butyrate are thought to be commonly implicated in sustaining the homeostasis of energy metabolism (Morrison and Preston, 2016). It has been identified that SCFAs profoundly exert some beneficial effects, including lowering blood glucose level, improving insulin sensitivity, and reducing inflammation, as well as elevating GLP-1 secretion (Feng et al., 2018; van der Beek et al., 2018; Wang et al., 2019; Zhang et al., 2018). Therefore, a counteractive effect eliciting by jicama fiber against HFD-induced T2DM development, as found in our present study, could be attributed to an increase in the SCFAs production and its subsequent implications in the blood glucose homeostasis. A further study examining the SCFA levels in both caeca and plasma under the supplementation of jicama fiber is needed to confirm our speculation.
The dynamic changes of gut microbiota composition have been reported to be closely associated with the development of metabolic disorders including those caused by HFD (Murphy et al., 2015; Peng et al., 2020). A previous study demonstrated that HFD could profoundly reduce the population of bacteria Akkermansia muciniphila, a key species promoting beneficial effects on host metabolism, in mice (He et al., 2018). Moreover, a study by Li et al. (2016) found that the reduction of gut microbiota diversity caused by HFD could be restored by the supplementation of fiber from the bamboo shoot in mice. Moreover, such restoration was also implicated in the improvement of metabolic outcomes including glycemic control and adiposity profiles. These findings suggest that the dietary fiber could also effectively counteract the development of metabolic diseases by modulating the gut microbiota of the host, thereby improvement of metabolic homeostasis. Although in our current study we did not analyze gut microbiota, we speculate that jicama fiber may also promote the counteractive effect against HFD-induced gut microbiota dysbiosis that could underlie its benefits in preventing T2DM. However, further investigation focusing on gut microbiota diversity of mice treated with jicama fiber should be carried out to clarify this speculation.
HFD is capable of promoting inflammation in tissues including insulin-targeted tissues (liver, adipose, and muscle) to cause insulin resistance as commonly observed in T2DM (Gao et al., 2015). Moreover, HFD also could induce pancreatic inflammation and subsequent degeneration of islet beta cells leading to insulin deficiency (Golson et al., 2010). Previous studies have indicated that jicama fiber exerts immunomodulatory effects both in vitro and in vivo particularly due to its pectin-like molecule as one of its active ingredients (Kumalasari et al., 2014a, 2014b). An anti-inflammatory effect of jicama fiber could also be mediated by inulin, a water-soluble fiber commonly found in jicama tuber. A study on patients with T2DM indicated that inulin supplementation could significantly reduce tumor necrosis factor-alpha in blood plasma, a marker of inflammation (Dehghan et al., 2014). Therefore, we also propose that jicama fiber might prevent the development of T2DM by eliciting its anti-inflammatory effect against HFD.
Although our present findings in the mice suggest a promising beneficial effect of consuming jicama fiber, at least there are two major limitations needed to be considered for future study. First, our present study used a relatively limited range of doses of jicama fiber (10% and 25%) that may preclude a proper justification on the best dose of jicama fiber with a maximum protective effect without or with fewer side effects against HFD. Second, the duration of treatment was relatively short (8 weeks). Consequently, it remains unanswered whether the beneficial effect of jicama fiber against the detrimental effects of HFD could stand for a longer period. Moreover, further investigations using pathological models as well as deploying molecular approaches are warranted to deepen our understanding of the mechanistic aspects of jicama fiber’s action.
CONCLUSION
The dietary fiber of jicama at the dose of 10% and 25% effectively prevented dysregulated blood glucose level and histopathological alterations of the pancreas in mice fed with HFD. Hence, jicama fiber could be considered as a potent supplement to preclude the development of T2DM due to excessive consumption of HFD.
AUTHORS’ CONTRIBUTIONS
PS and RM designed the study; PS, SJM, and QF performed the experiments; PS, RM, and RR analyzed the data and prepared the manuscript. All the authors have seen, and reviewed the manuscript and approved its final publication.
ACKNOWLEDGMENTS
This research was financially supported by the Indonesian Ministry of Research and Technology/National Agency for Research and Innovation (RISTEK/BRIN, Basic Research Grant No.163/SP2H/AMD/LT/DRPM/2020).
CONFLICT OF INTEREST
There were no conflicts of interest.
REFERENCES
American Diabetes Association. II: Classification and diagnosis of diabetes: standards of medical care in diabetes – 2019. Diabetes Care, 2019; 42(Suppl.1):13–28; doi:10.2337/dc19-S002 CrossRef
Bodnaruc AM, Prud’homme D, Blanchet R, Giroux I. Nutritional modulation of endogenous glucagon-like peptide-1 secretion: a review. Nutr Metab (Lond), 2016; 13:92; doi: 10.1186/s12986-016-0153-3 CrossRef
Buckman ES, Oduro I, Plahar WA, Tortoe C. Determination of the chemical and functional properties of yam bean (Pachyrhizus erosus (L.) Urban flour for food systems. Food Sci Nutr, 2017; 6:457–63; doi:10.1002/ fsn3.574 CrossRef
Burhans MS, Hagman DK, Kuzma JN, Schmidt KA, Kratz M. Contribution of adipose tissue inflammation to the development of type 2 diabetes mellitus. Compr Physiol, 2018; 9(1):1–58; doi:10.1002/cphy.c170040 CrossRef
Cantero I, Abete I, Monreal JI, Martinez JA, Zulet MA. Fruit fiber consumption specifically improves liver health status in obese subjects under energy restriction. Nutrients, 2017; 9(7):667; doi: 10.3390/nu9070667 CrossRef
de Munter JS, Hu FB, Spiegelman D, Franz M, van Dam RM. Whole grain, bran, and germ intake and risk of type 2 diabetes: a prospective cohort study and systematic review. PLoS Med, 2007; 4(8):e261.
Dehghan P, Gargari BP, Jafar-Abadi MA, Aliasgharzadeh A. Inulin controls inflammation and metabolic endotoxemia in women with type 2 diabetes mellitus: a randomized-controlled clinical trial, Int J Food Sci Nutr, 2014; 65(1):117–23; doi: 10.3109/09637486.2013.836738 CrossRef
Dhingra D, Michael M, Rajput H, Patil RT. Dietary fibre in foods: a review. J Food Sci Technol, 2012; 49(3):255–66; doi:10.1007/ s13197-011-0365-5 CrossRef
Eizirik DL, Pasquali L, Cnop M. Pancreatic β-cells in type 1 and type 2 diabetes mellitus: different pathways to failure. Nat Rev Endocrinol, 2020; 16:349–62; doi:10.1038/s41574-020-0355-7 CrossRef
Elkotby D, Hassan AK, Emad R, Bahgat I. Histological changes in islets of langerhans of pancreas in alloxan,induced diabetic rats following egyptian honey bee venom treatments. Int J Pure Appl Zool, 2017; 6(1):1–6.
Estrany ME, Proenza AM, Lladó I, Gianotti M. Isocaloric intake of a high-fat diet modifies adiposity and lipid handling in a sex dependent manner in rats. Lipids Health Dis, 2011; 10:52.
Feng W, Ao H, Peng C. Gut microbiota, short, chain fatty acids, and herbal medicines. Front Pharmacol, 2018; 9:1354; doi:10.3389/ fphar.2018.01354 CrossRef
Gao M, Ma Y, Liu D. High-fat diet-induced adiposity, adipose inflammation, hepatic steatosis and hyperinsulinemia in outbred CD-1 mice. PLoS One, 2015; 10(3):e0119784; doi:10.1371/journal.pone.0119784 CrossRef
Golson ML, Misfeldt AA, Kopsombut UG, Petersen CP, Gannon M. High fat diet regulation of β-cell proliferation and β-Cell mass. Open Endocrinol J, 2010; 4: 66-77; doi:10.2174/1874216501004010066 CrossRef
Guo W, Shu Y, Yang X. Tea dietary fiber improves serum and hepatic lipid profiles in mice fed a high cholesterol diet. Plant Foods Hum Nutr, 2016; 71(2):145–50; doi:10.1007/s11130-016-0536-7 CrossRef
Han S, Zhang W, Zhang R, Jiao J, Fu C, Tong X, Zhang W, Qin L. Cereal fiber improves blood cholesterol profiles and modulates intestinal cholesterol metabolism in C57BL/6 mice fed a high-fat- high-cholesterol diet. Food Nutr Res, 2019; 63:1-9; doi:10.29219/fnr.v63.1591 CrossRef
He C, Cheng D, Peng C, Li Y, Zhu Y, Lu N. High-fat diet induces dysbiosis of gastric microbiota prior to gut microbiota in association with metabolic disorders in mice. Front Microbiol, 2018; 9:639; doi:10.3389/ fmicb.2018.00639 CrossRef
Kumalasari ID, Nishi K, Harmayani E, Raharjo S, Sugahara T. Immunomodulatory activity of bengkoang (Pachyrhizus erosus) fiber extract in vitro and in vivo. Cytotechnology, 2014a; 66(1):75–85; doi:10.1007/s10616-013-9539-5 CrossRef
Kumalasari ID, Nishi K, Putra AB, Sugahara T. Activation of macrophages stimulated by the bengkoang fiber extract through toll-like receptor 4. Food Funct, 2014b; 5(7):1403–8. doi: 10.1039/c3fo60360a CrossRef
Larraufie P, Roberts GP, McGavigan AK, Kay RG, Li J, Leiter A, Melvin A, Biggs EK, Ravn P, Davy K, Hornigold DC, Yeo GSH, Hardwick RH, Reimann F, Gribble FM. Important role of the GLP-1 axis for glucose homeostasis after bariatric surgery. Cell Rep, 2019; 26(6):1399–408.e6; doi:10.1016/j.celrep.2019.01.047 CrossRef
Li X, Guo J, Ji K, Zhang P. Bamboo shoot fiber prevents obesity in mice by modulating the gut microbiota. Sci Rep, 2016; 6:32953; doi:10.1038/srep32953 CrossRef
Long Z, Zhang C, Sun Q, Liu Y, Liao N, Wu H, Wang X, Hai C. Evolution of metabolic disorder in rats fed high sucrose or high fat diet: focus on redox state and mitochondrial function. Gen Comp Endocrinol, 2017; 242:92–100; doi:10.1016/j.ygcen.2015.10.012 CrossRef
Lozano I, van der Werf R, Bietiger W, Seyfritz E, Peronet C, Pinget M, Jeandidier N, Maillard E, Marchioni E, Sigrist S, Dal S. High-fructose and high-fat diet-induced disorders in rats: impact on diabetes risk, hepatic and vascular complications. Nutr Metab, 2017; 13:15; doi:10.1186/ s12986-016-0074-1 CrossRef
Matsuda A, Makino N, Tozawa T, Shirahata N, Honda T, Ikeda Y, Sato H, Ito M, Kakizaki Y, Akamatsu M, Ueno Y, Kawata S. Pancreatic fat accumulation, fibrosis, and acinar cell injury in the zucker diabetic fatty rat fed a chronic high-fat diet. Pancreas, 2014; 43(5):735–43; doi:10.1097/ MPA.0000000000000129 CrossRef
Morrison DJ, Preston T. Formation of short chain fatty acids by the gut microbiota and their impact on human metabolism. Gut Microbes, 2016; 7(3):189–200; doi:10.1080/19490976.2015.1134082 CrossRef
Murphy EA, Velazquez KT, Herbert KM. Influence of high-fat diet on gut microbiota: a driving force for chronic disease risk. Curr Opin Clin Nutr Metab Care, 2015; 18(5):515–20; doi:10.1097/ MCO.0000000000000209 CrossRef
Nadkarni P, Chepurny OG, Holz GG. Regulation of glucose homeostasis by GLP-1. Prog Mol Biol Transl Sci, 2014; 121:23–65; doi:10.1016/B978-0-12-800101-1.00002-8 CrossRef
Nagy C, EinWallner E. Study of in vivo glucose metabolism in high, fat diet, fed mice using oral glucose tolerance test (ogtt) and insulin tolerance test (ITT). J Vis Exp, 2018; 131:e56672; doi:10.3791/56672 CrossRef
Noman ASM, Hoque MA, Haque MM, Pervin F, Karim MR. Nutritional and anti-nutritional components of Pachyrhizus erosus L. tuber. Food Chem, 2012; 102:1112–8; doi:10.1016/j.foodchem.2006.06.055 CrossRef
Park CJ, Han JS. Hypoglycemic effect of jicama (Pachyrhizus erosus) extract on streptozotocin, induced diabetic mice. Prev Nutr Food Sci, 2015; 20(2):88–93; doi:10.3746/pnf.2015.20.2.88 CrossRef
Park CJ, Lee HA, Han JS. Jicama (Pachyrhizus erosus) extract increases insulin sensitivity and regulates hepatic glucose in C57BL/Ksj-db/ db mice. J Clin Biochem Nutr, 2016; 58(1):56–63; doi:10.3164/jcbn.15-59 CrossRef
Patel H, Patel VH. Inflammation and metabolic syndrome, an overview. Curr Res Nutr Food Sci, 2015; 3(3):263-268; doi:10.12944/ CRNFSJ.3.3.10 CrossRef
Pearson ER. Type 2 diabetes: a multifaceted disease. Diabetologia, 2019; 62:1107–112; doi:10.1007/s00125-019-4909-y CrossRef
Peng C, Xu X, Li Y, Li X, Yang X, Chen H, Zhu Y, Lu N, He C. Sex-specific association between the gut microbiome and high-fat diet-induced metabolic disorders in mice. Biol Sex Differ, 2020; 11(1):5; doi:10.1186/s13293-020-0281-3 CrossRef
Pouyamanesh Z, Amoli MM, Yaghmaei P, Ebrahim-Habibi A. Effect of inulin supplementation in male mice fed with high fat diet on biochemical profile and α-amylase gene expression. Trop J Pharm Res, 2016; 15(6):1197–203; doi:10.1016/S0016-5085(17)33059-7 CrossRef
Puddu A, Sanguineti R, Montecucco F, Viviani GL. Evidence for the gut microbiota short-chain fatty acids as key pathophysiological molecules improving diabetes. Mediators Inflamm, 2014; 2014:162021; doi:10.1155/2014/162021 CrossRef
Saklayen MG. The global epidemic of the metabolic syndrome. Curr Hypertens Rep, 2019; 20(2):12; doi:10.1007/s11906-018-0812-z CrossRef
Santoso P, Amelia A, Rahayu R. Jicama (Pachyrhizus erosus) fiber prevents excessive blood glucose and body weight increase without affecting food intake in mice fed with high,sugar diet. J Adv Vet Anim Res, 2019; 6(2):222–30; doi:10.5455/javar.2019.f336 CrossRef
Santoso P, Maliza R, Fadhilah Q, Insani SJ. Beneficial effect of Pachyrhizus erosus fiber as a supplemental diet to counteract high sugar-induced fatty liver disease in mice. Rom J Diabetes Nutr Metab Dis, 2020a; 26(4):353–60; doi:10.2478/rjdnmd-2019-0038 CrossRef
Santoso P, Maliza R, Rahayu R, Amelia A. Pancreoprotective effect of jicama (Pachyrhizus erosus, Fabaceae) fiber against high, sugar diet in mice. Open Access Maced J Med Sci, 2020b; 8(A):1–7; doi:10.3889/ oamjms.2020.4528 CrossRef
Skovsø S. Modeling type 2 diabetes in rats using high fat diet and streptozotocin. J Diabetes Investig, 2014; 5(4):349–58; doi:10.1111/ jdi.12235 CrossRef
Thaptimthong T, Kasemsuk T, Sibmooh N, Unchern S. Platelet inhibitory effects of juices from pachyrhizus erosus L. root and Psidium guajava L. fruit: a randomized controlled trial in healthy volunteers. BMC Complement Altern Med, 2016; 16:269; doi:10.1186/s12906-016-1255-1 CrossRef
van der Beek CM, Canfora EE, Kip AM, Gorissen SHM, Damink SWMO, ven Eijk HM, Holst JJ, Blaak EE, Dejong CHC, Lenaerts K. The prebiotic inulin improves substrate metabolism and promotes short chain fatty acid production in overweight to obese men. Metab Clin Exp, 2018; 87:25–35; doi:10.1016/j.metabol.2018.06.009 CrossRef
Wang D, Liu CD, Tian ML, Tan CQ, Shu G, Jiang QY, Zhang L, Yin Y. Propionate promotes intestinal lipolysis and metabolic benefits via AMPK/LSD1 pathway in mice. J Endocrinol, 2019; 19:0188. doi:10.1530/ JOE-19-0188 CrossRef
Wang ZQ, Yu Y, Zhang XH, Floyd EZ, Bourdreau A, Lian K, Cefalu WT. Comparing the effects of nano-sized sugarcane fiber with cellulose and psyllium on hepatic cellular signaling in mice. Int J Nanomedicine, 2012; 7:2999–3012; doi:10.2147/IJN.S30887 CrossRef
Ye T, Chen YH, Gao JH, Wang XX, Qiang O, Tang CW, Liu R. Effect of octreotide on pancreatic fibrosis in rats with high-fat diet-induced obesity. Int J Clin Exp Pathol, 2018; 11(10):4784–94.
Yu T, Liu R, Li M, Li X, Qiang O, Huang W, Tang C. Effects of octreotide on fatty infiltration of the pancreas in high-fat diet induced obesity rats. Wei Sheng Yan Jiu, 2014; 43(2):186–92.
Zaharia OP, Strassburger K, Strom A, Bönhof GJ, Karusheva Y, Antoniou S, Bódis K, Markgraf DF, Burkart V, Müssig K, Hwang JH, Asplund O, Groop L, Ahlqvist E, Seissler J, Nawroth P, Kopf S, Schmid SM, Stumvoll M, Pfeiffer AFH, Kabisch S, Tselmin S, Häring HU, Ziegler D, Kuss O, Szendroedi J, Roden M, German Diabetes Study Group. Risk of diabetes, associated diseases in subgroups of patients with recent-onset diabetes: a 5-year follow-up study. Lancet Diabetes Endocrinol, 2019; 7(9):684–94; doi:10.1016/S2213-8587(19)30187-1 CrossRef
Zhai X, Lin D, Zhao Y, Li W, Yang X. Effects of dietary fiber supplementation on fatty acid metabolism and intestinal microbiota diversity in C57BL/6J mice fed with a high-fat diet. J Agric Food Chem, 2018a; 66(48):12706–18; doi:10.1021/acs.jafc.8b05036 CrossRef
Zhai X, Lin D, Zhao Y, Li W, Yang X. Enhanced anti, obesity effects of bacterial cellulose combined with konjac glucomannan in high-fat diet-fed C57BL/6J mice. Food Funct, 2018b; 9(10):5260–72; doi:10.1039/ c8fo01211c CrossRef
Zhang Q, Yu H, Xiao X, Hu L, Xin F, Yu X. Inulin-type fructan improves diabetic phenotype and gut microbiota profiles in rats. PeerJ, 2018; 6:e4446; doi:10.7717/peerj.4446 CrossRef